Barcelona, Spain (UroToday.com) While the standard of care for non-metastatic muscle-invasive bladder cancer remains neoadjuvant platinum-based chemotherapy followed by radical cystectomy, a number of recently completed and ongoing clinical trials are investigating the use of checkpoint blockade immunotherapy in this setting. Immunotherapy is an attractive neoadjuvant option given its very good safety profile and tolerability, especially among cisplatin-ineligible patients. NABUCCO is a phase Ib single arm trial of neoadjuvant combination checkpoint blockade immunotherapy with the CTLA-4 antibody ipilimumab and the PD-1 antibody nivolumab among cisplatin-ineligible patients with high-risk stage III urothelial carcinoma.
Patients were required to have cT3-4aN0M0 or ≥cT1-4aN1-3 disease at entry and to be ineligible to receive platinum-based chemotherapy. Of course, patients with distant lymph node metastases were excluded, as were all other stage IV patients. Investigators utilized a sequential dosing regimen with ipilimumab (3mg/kg on day 1) followed by ipilimumab plus nivolumab (1 mg/kg on day 22), and nivolumab alone (day 43). Per protocol, all patients were planned to undergo surgical resection and lymphadenectomy between days 57 and 71. The primary endpoint was feasibility as defined by the rate of surgical resection within 12 weeks. Key secondary endpoints were efficacy (including pathologic complete response rate) and toxicity.
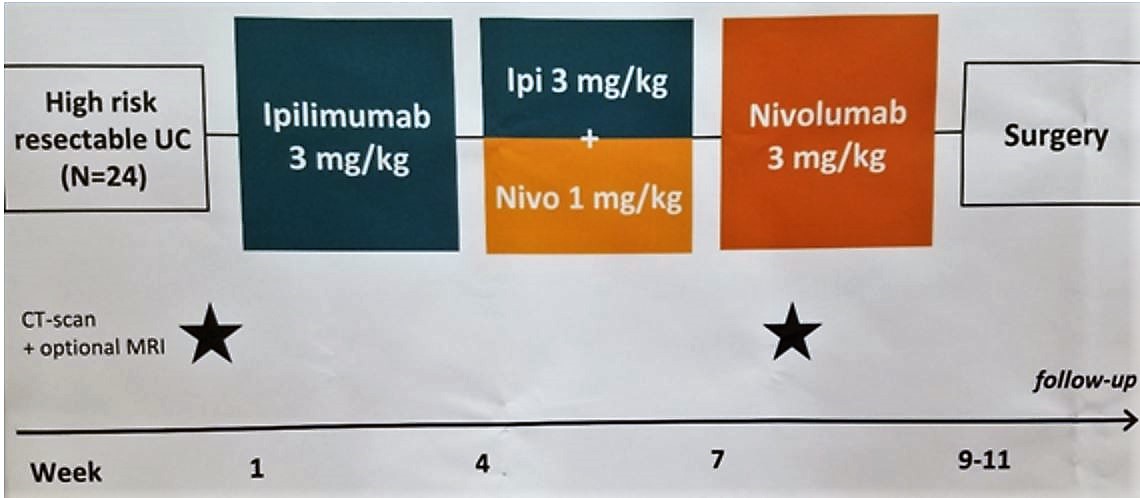
A total of 24 patients were treated on this neoadjuvant protocol, with a mean age of 65 and a 75% male predominance. While the majority 58% of patients did not have nodal disease (cT3-T4aN0M0), 38% of patients did have nodal disease (cT1-4N1-3M0) and a single patient (4%) was restaged after enrollment to cT4bN1M0. Regarding PD-L1 status, 63% of patients had a combined positive score ≥10%.

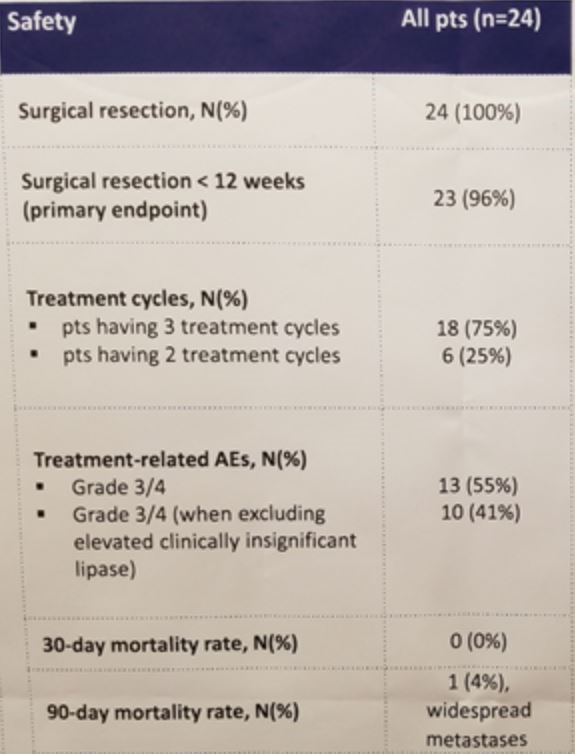
With respect to the primary endpoint, 100% of patients ultimately underwent a radical cystectomy (or nephroureterectomy in the case of a single upper tract patient); only a single patient was delayed beyond the 12-week timeframe established by the protocol for toxicity (hemolysis). Not unexpectedly, a majority of patients (55%) did experience some grade 3 or grade 4 toxicity, though it is reassuring that all were able to undergo surgical resection and no patient died within the 30-day postoperative window. No new or unexpected safety signals were observed with ipilimuam plus nivolumab at the prescribed dosing levels. However, 25% of patients were only able to receive 2 of 3 planned cycles, and a single patient died within 3 months of surgery due to widespread metastatic disease.
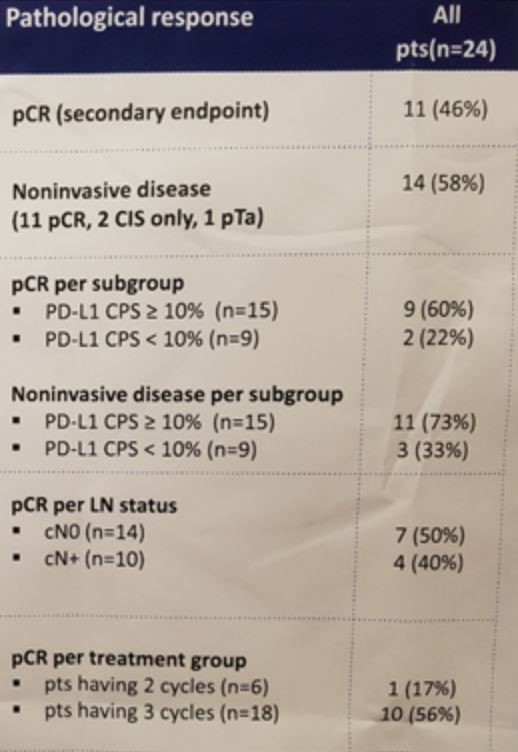
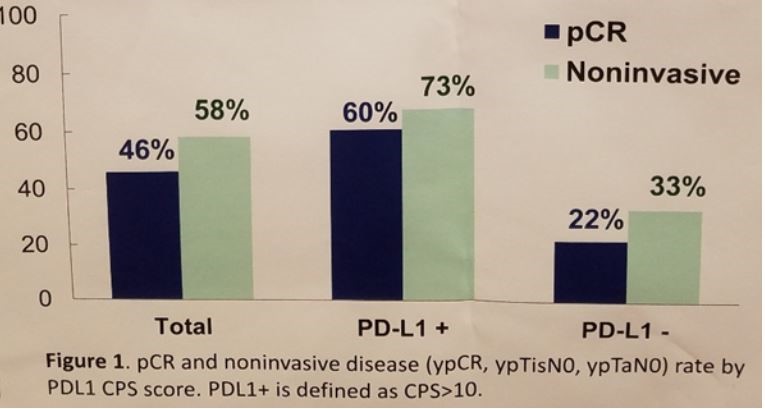
A total of 46% of the entire cohort achieved a pathologic complete response, a remarkable outcome given the high-risk nature of these locoregionally-advanced tumors. Furthermore, a total of 58% were downstaged to non-invasive disease at the time of surgery. Even 40% of the patients with nodal disease achieved a pCR. PD-L1 status appeared to enrich for responses, with 60% achieving a pCR in the CPS ≥10% cohort relative to just 22% in the PD-L1 low cohort.
In summary, neoadjuvant ipilimumab with nivolumab was feasible for high-risk stage III urothelial carcinoma patients. While NABUCCO was a small trial, not a single of the 24 enrolled patients progressed while on treatment, and all were able to have undergo definitive surgical resection with impressive pathologic complete response rates. Follow-up cohorts will examine the use of flip-dose ipilimuam (1 mg/kg) with nivolumab (3 mg/kg).
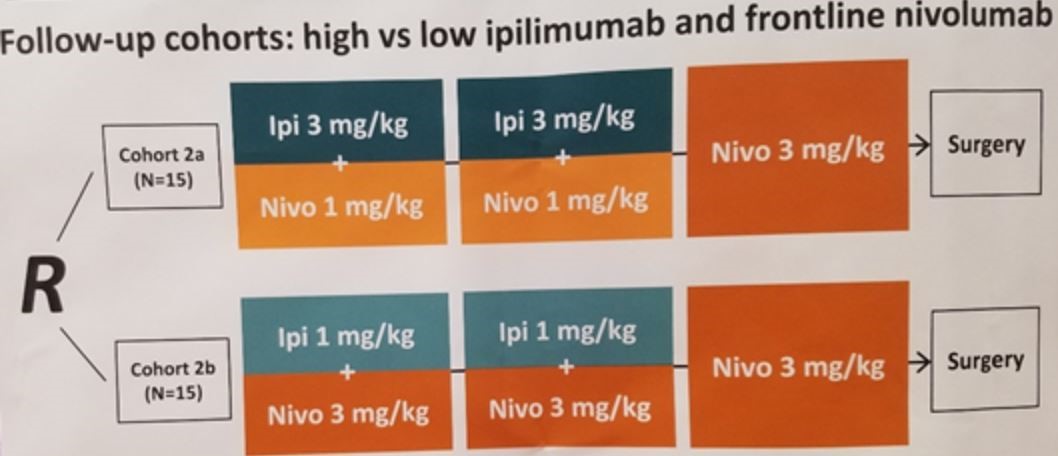
Longer follow-up and larger trials will be required to validate these promising findings and establish benefit with respect to recurrence-free and overall survival.
Presented by: Michiel S. Van der Heijden, MD, PhD, Netherlands Cancer Institute, Amsterdam, Netherlands
Written by: Michael Lattanzi, MD, Medical Oncology Fellow, Memorial Sloan Kettering Cancer Center, Twitter: @MikeLattanzi at the 2019 European Society for Medical Oncology Congress (#ESMO19), September 27 – October 1, 2019, Barcelona, Spain
