Basel, Switzerland (UroToday.com) Dr. Darren Poon gave an overview of prostate cancer in the East-Asian ethnicity. This ethnicity includes China, (including Hong-Kong and Macau), Japan, Mongolia, North Korea, South Korea, and Taiwan.
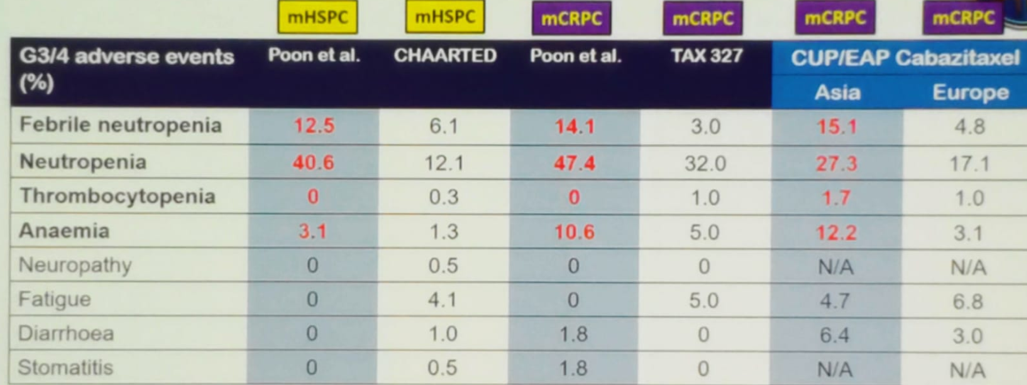
When assessing the taxane related adverse events in Asian prostate cancer patients, compared to the rates described in the major studies, there is clear evidence showing that Asian people have a higher rate of adverse events, as can be seen in table 1. It appears that Asian people are more susceptible to docetaxel’s myelosuppression (with an odds ratio of 19, 95% CI 3.64-99.0, p<0.001).
Table 1 – Taxane related adverse events in Asian prostate cancer patients compared to the rates described in the corresponding landmark trials:
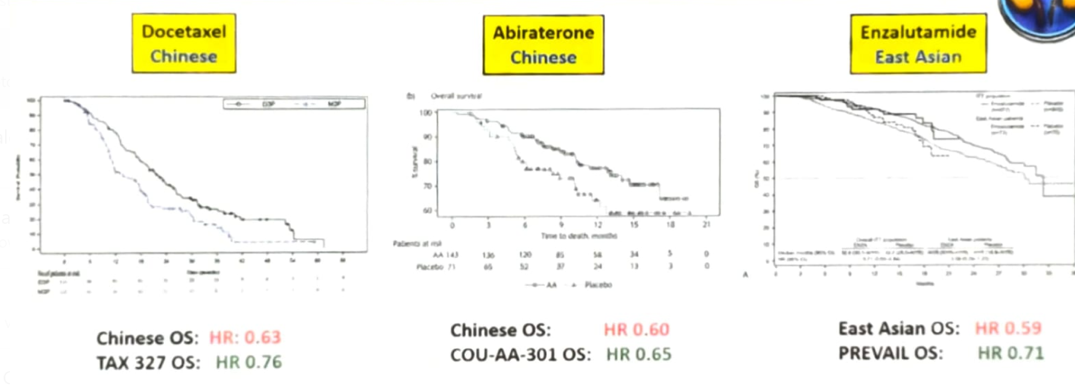
Interestingly, the differences in adverse events are not exclusive to taxanes, and even in androgen-signaling pathway inhibitors, there is a difference in the rate of adverse events when comparing global rates to that of Asian people (Table 2). However, when assessing the treatment efficacy, it would seem to be similar between Asian and Caucasian people, for enzalutamide, abiraterone, and docetaxel (Figure 1).
Table 2 – Adverse events in androgen signaling pathway inhibitors in Asians and Global data:
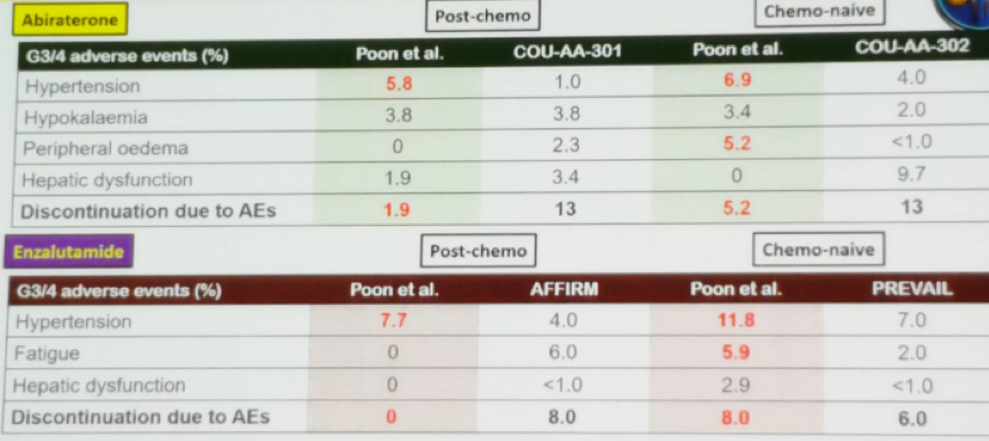
Figure 1 – Comparison of treatment efficacy between Asian and Caucasian people:
Docetaxel clearance is a significant predictor for febrile neutropenia. Asian people have smaller bodyweight than Caucasian men (an average of 20 kg). They have a smaller body build with a limited marrow reserve, that might explain the higher risk of taxane-related myelosuppression seen in the Asian population using the same docetaxel dose (Table 3). Possible solutions for this include docetaxel dose/schedule modification with reduction of the usual dose from 75 mg/m2 to 60 mg/m2. Another possibility is to alter the frequency from every 3 weeks to every 2 weeks. One other option is to use the supportive measure of preemptive granulocyte-colony stimulating factor (GCSF). According to the Hong-Kong consensus on prostate cancer management,1 these solutions should be utilized in order to reduce the rate of adverse effects. A recently published Japanese study showed that with the preemptive use of GCSF the febrile neutropenia rate caused by docetaxel in metastatic castrate-resistant prostate cancer patients decreased from 54% to 9%.2
Table 3 – Difference in body weight between Caucasian and Asian men: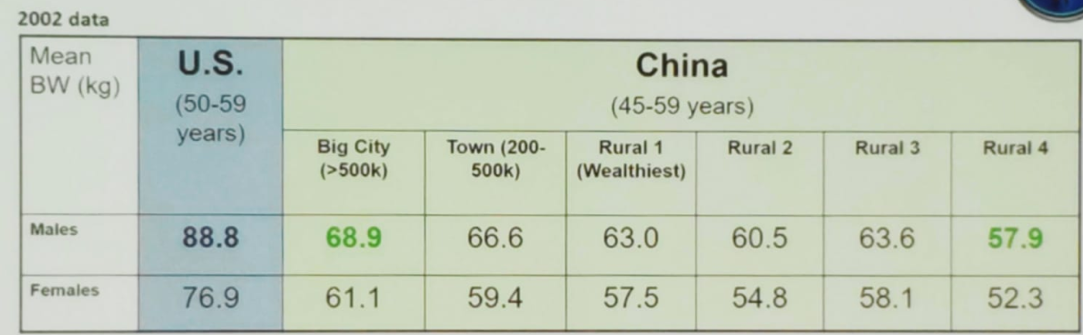
According to Dr. Poon, there is currently insufficient Asian contribution in clinical trials and fundamental research, creating disparities in data regarding Asian patients.
Summarizing what is currently known regarding Asian prostate cancer patients, including the fact that they have a higher risk of hematological complications with taxanes, which may be related to their smaller body build with limited marrow reserve. They do tolerate androgen receptor signaling pathway inhibitors quite well with no significant differences in survival outcomes when compared to the rest of the world. In an attempt to alleviate the risk of taxane-related myelosuppression in Asian prostate cancer patients, possible solutions include dose/schedule alterations and preemptive use of GCSF. Lastly, pharmaco-ethnicity should be considered in future trials and fundamental cancer research.
Presented by: Darren MC Poon, FHKCR, Department of Clinical Oncology, The Chinese University of Hong Kong, Hong Kong
Written by: Hanan Goldberg, MD, Urology Department, SUNY Upstate Medical University, Syracuse, New-York, USA @GoldbergHanan at the 2019 Advanced Prostate Cancer Consensus Conference (APCCC) #APCCC19, Aug 29 – 31, 2019 in Basel, Switzerland
References:
- Poon DMC et al. Survival Outcomes, Prostate-specific Antigen Response, and Tolerance in First and Later Lines of Enzalutamide Treatment for Metastatic Castration-resistant Prostate Cancer: A Real-World Experience in Hong Kong. BJU International. 2018 Oct;16(5):402-412.e1. doi: 10.1016/j.clgc.2018.07.008. Epub 2018 Jul 21.
- Kosaka T. et al. Impact of pegfilgrastim as primary prophylaxis for metastatic castration-resistant prostate cancer patients undergoing cabazitaxel treatment: an open-label study in Japan. Japanese Journal of Clinical Oncology. 2019 Apr 26. pii: hyz051. doi: 10.1093/jjco/hyz051.
