Basel, Switzerland (UroToday.com) Professor Anwar Padhani from the United Kingdom discussed imaging and flare responses when assessing patients with metastatic castration-resistant prostate cancer (mCRPC) at the Bone and Bone Metastases session at the 2019 Advanced Prostate Cancer Consensus Conference (APCCC2019). Dr. Padhani notes that bone scans/CT scans lead to poor confidence for knowing the true clinical states and assessing therapy benefits for bone disease in advanced prostate cancer with precision oncology implications. Conventional imaging tools are unable to accurately predict metastatic disease presence and extent, as well as being unable to identify who is not benefiting early after starting treatment. Dr. Padhani suggests that these limitations have several implications for precision oncology:
- Late detection and volume underestimation of metastatic disease (inaccurate tumor volume estimates and inaccurate risk allocations) misdirect therapy approaches
- Fail to rapidly detect primary and secondary resistance, impeding patient progression to alternative therapies, and subsequently increasing costs
- Heterogeneity of response is increasingly important for predicting patient outcomes
New bone scan lesions on the first follow-up scan may represent true progression, tumor ‘momentum’ (temporarily worsening disease before delayed ADT response), or early favorable osteoblastic reaction (ie. bone scan flare or pseudo progression). The Prostate Cancer Working Group has developed the 2+2 Rule: new focal bone scan lesions that develop in the flare period (8-12 weeks) should be confirmed with the documentation of additional new lesions on the next follow-up scan (in the absence of other signs of progression). The development of new focal bone lesions on the first follow-up bone scan (unconfirmed) in the absence of clinical progression, often represents a favorable response to treatment.
however there is PSA-discordant progression (clinical and imaging progression in the absence of a PSA rise) in 30-45% of men on ADT/docetaxel. For mCRPC, symptoms and PSA are commonly used for monitoring, but are increasingly considered inadequate. PSA-discordant progression occurs in 21-30% of men where they can develop radiographic progression without clinical/PSA progression on abiraterone and enzalutamide. The imaging flare is not limited to bone scan imaging but is also seen in NaF PET/CT, CT, PSMA PET/CT, MRI morphology, MRI diffusion.
Dr. Padhani notes that there are several strategies to overcoming the limitations of bone scan imaging flare, including functional imaging:

Dr. Padhani and his group have increasingly been utilizing whole body diffusion-weighted (DW)-MRI for imaging patients with mCRPC. Diffusion-weighted imaging allows spatial mapping and quantification of cellular viability habitats over a series of temporal images. He calls this the “traffic light assessment”: Alive (red), Dying (yellow), and Dead (green) disease:
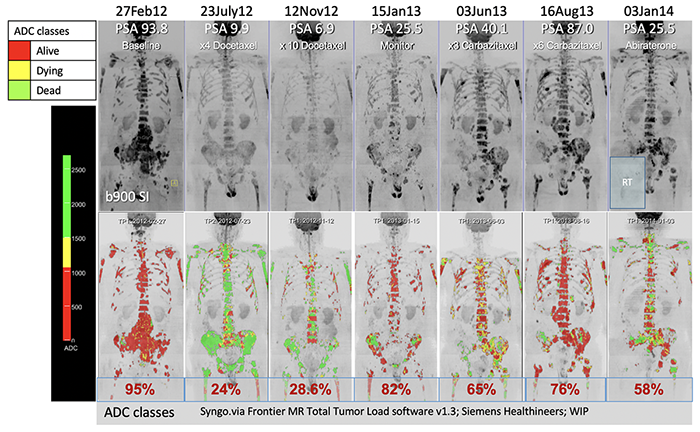
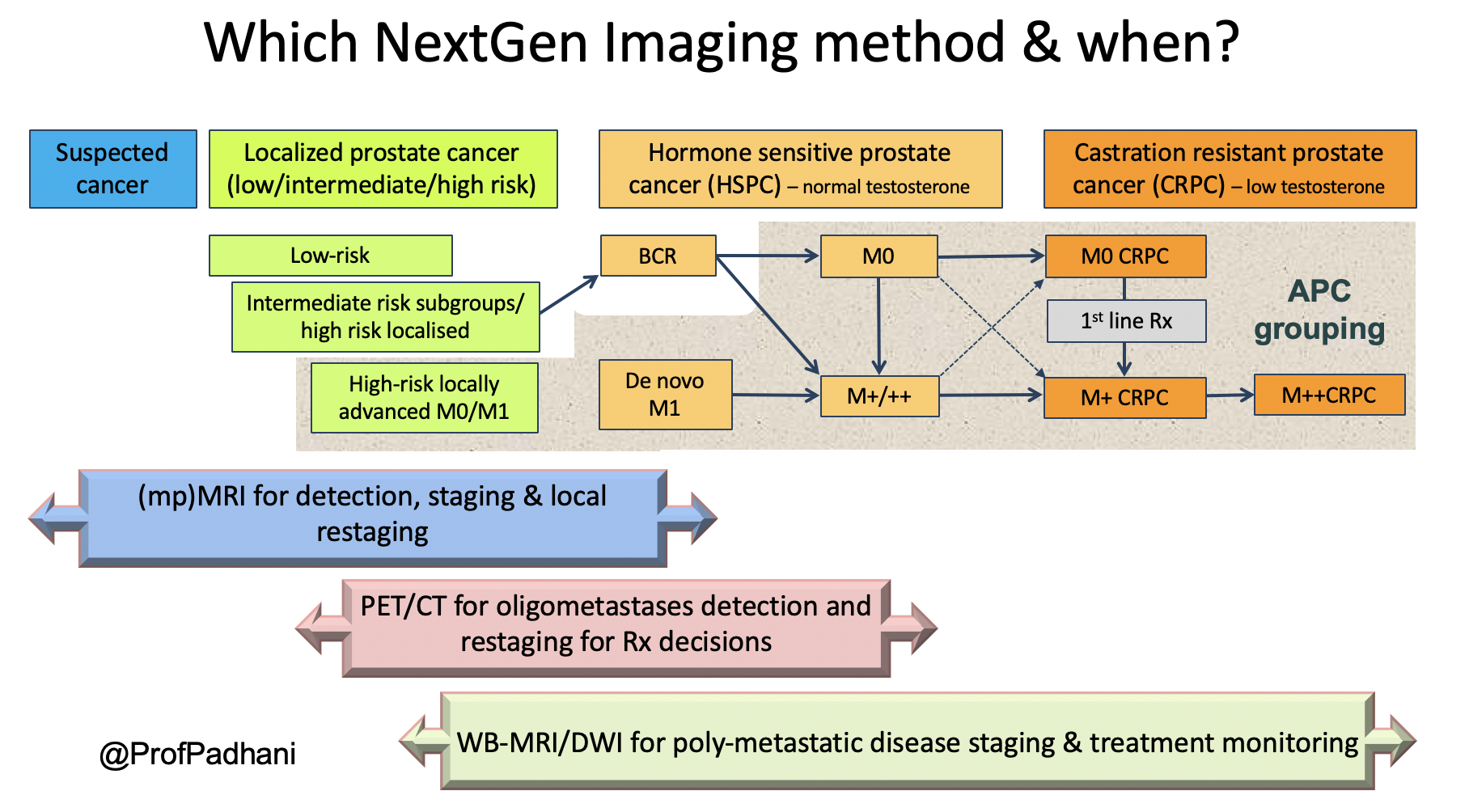
Dr. Padhani advocates that next generation imaging promotes precision oncology trials and that treatment response assessments allow for the right treatment, for the right patient, at the right time, for the right duration. This is summarized in the following figure assessing imaging modalities along the prostate cancer spectrum timeline:
Dr. Padhani concluded his presentation with several take home points, noting that it is time to move on to next-generation imaging and to stop relying on CT/bone scans for bone metastatic assessments:
- Next generation imaging PET-CT and whole-body MRI/DWI can detect bone metastases with high sensitivity – early detection of metastases and therapy resistance can have positive precision oncology therapy implications
- Next generation imaging can positively assess therapy response and early failure, unlike bone scans which only identify late tumor progression – accurate therapy assessments and heterogeneity can promote the development and clinical use of targeted therapies
- Next generation imaging directed therapy survival benefits in advanced prostate cancer have not been evaluated, but methods are ready for clinical trial incorporation
Presented by: Anwar R. Padhani, Mount Vernon Hospital, Northwood, England
Written by: Zachary Klaassen, MD, MSc – Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia Twitter: @zklaassen_md at the 2019 Advanced Prostate Cancer Consensus Conference (APCCC) #APCCC19, Aug 29 – 31, 2019 in Basel, Switzerland
