Basel, Switzerland (UroToday.com) Dr. Gleave gave a discussion on the topic of testosterone measurement and its various nuances in the context of advanced prostate cancer.
The discovery of how testosterone is synthesized resulted in the receipt of the Nobel prize in 1939 by Adam Butenandt and Leopold Ruzicka. Another Nobel prize was awarded to Charles Huggins in 1966 for discoveries concerning the hormonal treatment of prostate cancer.
Testosterone measurement is important as androgens are the drivers of activity of the androgen receptor (AR). Maintenance of castrate testosterone levels required in metastatic castrate-resistant prostate cancer (mCRPC) is a key factor for intermittent and continuous androgen deprivation therapy (ADT). By consensus, testosterone levels below 50 ng/dl (1.7 nM) is defined as castrate. It is also known that serum testosterone levels (and perhaps other serum androgens) is prognostic for time to CRPC and potentially predictive of AR signaling pathway inhibition.
It is important to note that some patients on ADT will not achieve castrate testosterone levels. At a testosterone level of 1.7 nmol/l approximately 63-87% will achieve castration1-4, while at a testosterone level of 0.7 nmol/l, approximately 88-97% will achieve castration1,2,4,5. The combination of LHRH agonists with abiraterone will usually achieve testosterone levels of less than 0.3 nM. Time to CRPC on LHRH agonists has been linked to serum testosterone levels6.7.
In the COU-301 trial8 serum testosterone levels in mCRPC patients were measured by LC-MS/MS and were shown to be prognostic for overall survival (Figure 1), with longer overall survival rates in patients with serum androgens above the median.
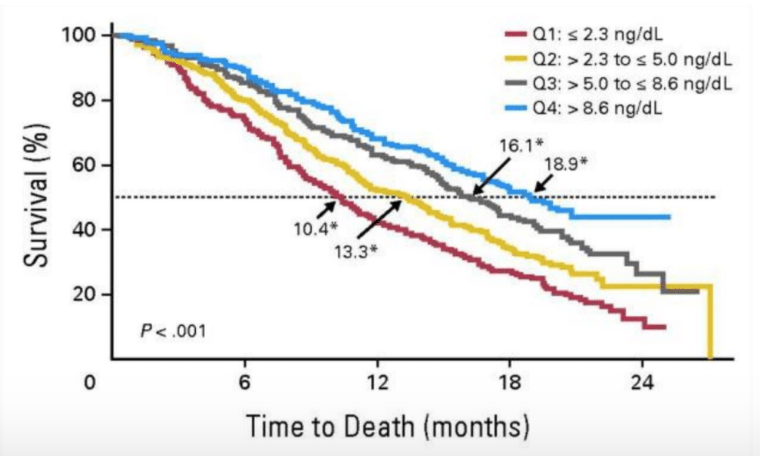
Figure 1. Longer overall survival in patients with serum androgens above the median (p<0.0001) in the Cou-301 trial
The quantification of testosterone at low levels in men on ADT is challenging as it is the limit of quantification. Historically, testosterone was measured with solvent extraction followed by RIA or GC-MS. Automated immunoassays for total testosterone were developed for high-throughput, clinical laboratory environment. The automated immunoassays are unfortunately inaccurate at low testosterone levels due to endogenous and exogenous interferences, and issues with calibration at low analyte concentrations. Approximately 3% of samples with testosterone levels below 0.7 nM by automated immunoassay were actually above 0.7 nM, using the LC-MS method. Therefore, suboptimal testosterone levels in castrated patients should be confirmed by either MS or a validated immunoassay and interpreted with caution before any changes are made to treatment.
The Canadian consensus statement recommends achieving a serum testosterone level of less than 0.7nmol/l as there appears to be an associated clinical benefit9. Additional recommendations include regular monitoring of testosterone and PSA levels throughout the first year of ADT treatment, using validated LC-MS/MS methods for testosterone assays at low testosterone levels (<0.7 nmol/l).
In conclusion, lower testosterone levels correlate with better freedom from CRPC. Breakthrough testosterone levels above 1.7 nmol/l have higher rates of failure. Testosterone levels should be assessed periodically to ensure castrate levels (every 3- months), with confirmation of lab assays at low testosterone levels. Lastly, the role of testosterone and other serum androgens as prognostic and predictive biomarkers in mCRPC requires more study.
Presented by: Dr. Martin Gleave, Vancouver, Canada
References:
1. Oefelein MG, Cornum R. Failure to achieve castrate levels of testosterone during luteinizing hormone releasing hormone agonist therapy: the case for monitoring serum testosterone and a treatment decision algorithm. The Journal of urology. Sep 2000;164(3 Pt 1):726-729.
2. Morote J, Esquena S, Abascal JM, et al. Failure to maintain a suppressed level of serum testosterone during long-acting depot luteinizing hormone-releasing hormone agonist therapy in patients with advanced prostate cancer. Urologia internationalis. 2006;77(2):135-138.
3. Wechsel HW, Zerbib M, Pagano F, Coptcoat MJ. Randomized open labelled comparative study of the efficacy, safety and tolerability of leuprorelin acetate 1M and 3M depot in patients with advanced prostatic cancer. European urology. 1996;30 Suppl 1:7-14; discussion 19-21.
4. McLeod, David, Norman Zinner, Kevin Tomera, Donald Gleason, Nick Fotheringham, Marilyn Campion, Marc B. Garnick, and Abarelix Study Group. “A phase 3, multicenter, open-label, randomized study of abarelix versus leuprolide acetate in men with prostate cancer.” Urology 58, no. 5 (2001): 756-761.
5. Kawakami J, Morales A. Clinical significance of suboptimal hormonal levels in men with prostate cancer treated with LHRH agonists. Canadian Urological Association journal = Journal de l’Association des urologues du Canada. Mar-Apr 2013;7(3-4):E226-230.
6. lotz L, O’Callaghan C, Ding K, et al. Nadir. Testosterone within first year of androgen-deprivation therapy (ADT) predicts for time to castration-resistant progression: a secondary analysis of the PR-7 trial of intermittent versus continuous ADT. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. Apr 1 2015;33(10):1151-1156.
8. Ryan CJ, Molina A, Li J, et al. Serum androgens as prognostic biomarkers in castration-resistant prostate cancer: results from an analysis of a randomized phase III trial. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. Aug 1 2013;31(22):2791-2798.
9. Klotz L, Shayegan B, Guillemette C, et al. Testosterone suppression in the treatment of recurrent or metastatic prostate cancer – A Canadian consensus statement. Canadian Urological Association journal = Journal de l’Association des urologues du Canada. Feb 2018;12(2):30-37.
