Basel, Switzerland (UroToday.com) Dr. Shore gave a talk on the metabolic complications of androgen deprivation therapy (ADT) and how to manage them. ADT has many complications, including skeletal events, cardiovascular disease, diabetes, neurocognitive disease, and others. ADT also confers significant quality of life complications including hot flashes, loss of libido, erectile dysfunction, fatigue, and others.
Dr. Shore focused on the cardiovascular complications caused by ADT. Androgens have a protective role in the cardiovascular system. They cause coronary vasodilation, maintain plaque stability, have an anti-arrhythmic effect and shorten the QT segment. Additionally, they have a favorable effect on body composition. In men with coronary artery disease, low testosterone level is a marker of increased all-cause and cardiovascular mortality1.
Next, Dr. Shore discussed the association of testosterone and ADT with obesity. Low testosterone levels have been correlated with obesity. The average weight gain after a year of ADT is approximately 1.5-4 kg and greater in younger and non-obese patients. There is also an increase in body fat of up to 9-11% and a decrease in lean body mass of 3-4%. The obesity that usually occurs is described as sarcopenic obesity which is abdominal obesity and reduced muscle mass, frequently associated with an increase in all-cause mortality.
Dr. Shore continued and described the association of dyslipidemia and ADT. ADT has been shown to increase triglyceride levels (in approximately 26% of patients), total cholesterol levels (in approximately 10% of patients), LDL levels (7%), and HDL (8-10%). Despite the increase of HDL, the low testosterone levels are associated with a lack of uptake of cholesterol from the arterial wall to the liver, culminating in a net proatherogenic effect.
Metabolic syndrome is prevalent in approximately 20-25% of the adult population. Male hypogonadism is an independent risk factor for metabolic syndrome, while low testosterone and serum hormone-binding globulin (SHBG) levels can predict metabolic syndrome in men. Approximately 35-65% of men receiving ADT for more than 1 year develop metabolic syndrome independent of age, race, and stage of prostate cancer2.
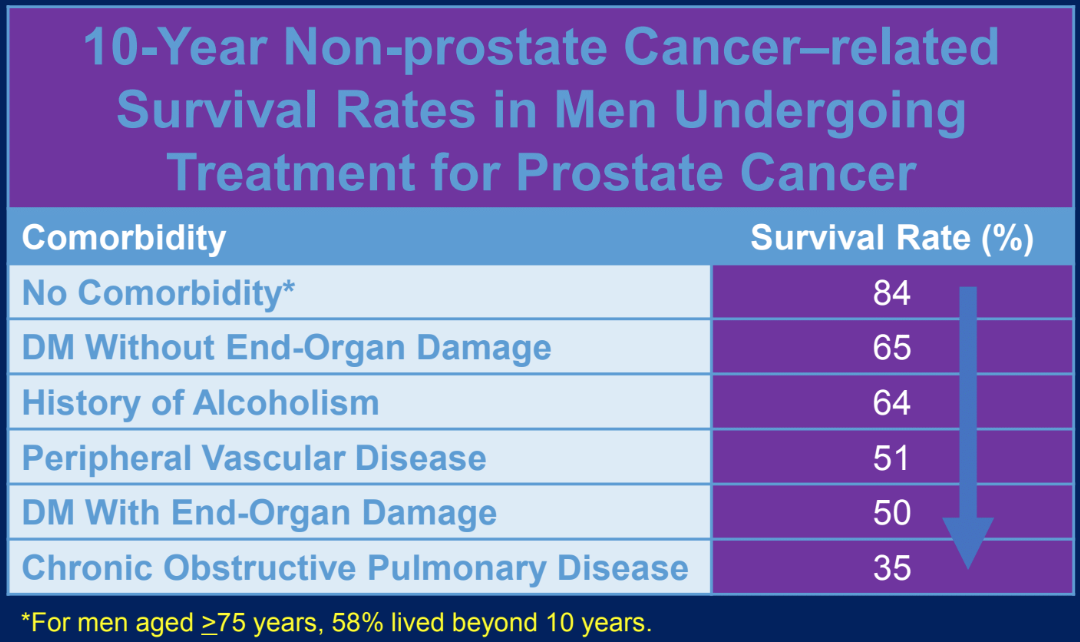
The comorbidities that are developed while being treated with ADT ultimately affect survival (Table 1). GnRH agonists have been shown to increase the risk of cardiovascular disease in 21% of men3. The risk of cardiovascular disease is highest in the first 6 months of ADT in men who experienced 2 or more cardiovascular events within the year prior to therapy. However, the current literature on ADT and cardiovascular risk is inconsistent. The randomized trials in the past did not include higher cardiac risk elderly patients and hence were underpowered to assess cardiovascular outcomes. Additionally, the cardiovascular outcomes were not studied as a primary endpoint. Not surprisingly, secondary analysis from these trials has failed to show an association between ADT and adverse cardiovascular sequelae, a finding that has been consistently shown in large-scale observational studies.
Table 1. Comorbidities impact survival among men with prostate cancer
According to the National Comprehensive Cancer Network (NCCN) guidelines in 2018 for cardiovascular risk in men on ADT, it is critical to assess traditional risk factors for cardiovascular disease using the ABCDE approach (Table 2). Moreover, this should be a multidisciplinary effort involving the primary care physician, the geriatrician, and the cardio-oncologist or cardiologist.
Table 2. ABCDE approach for cardiovascular risk assessment for men on ADT

The FDA issued a warning in 2010 for GnRH agonists harboring an increased risk for cardiovascular disease namely coronary heart disease, myocardial infarction, and sudden cardiac death. It is currently not clear if the LHRH antagonists harbor a lower risk than the agonists. This resulted in the creation of the PRONOUNCE trial which randomizes patients referred for ADT to be treated with either degarelix (LHRH antagonist) or leuprolide (LHRH agonist). The number of the all the accrued patients is 900 patients with predefined cardiac disease. The study’s primary endpoint is time from randomization to first confirmed occurrence of a composite major adverse event (MACE). MACE is defined as death from any cause, non-fatal myocardial infarction, and non-fatal stroke. Figure 1 demonstrates an algorithm developed to address treatment for prostate cancer treatment.
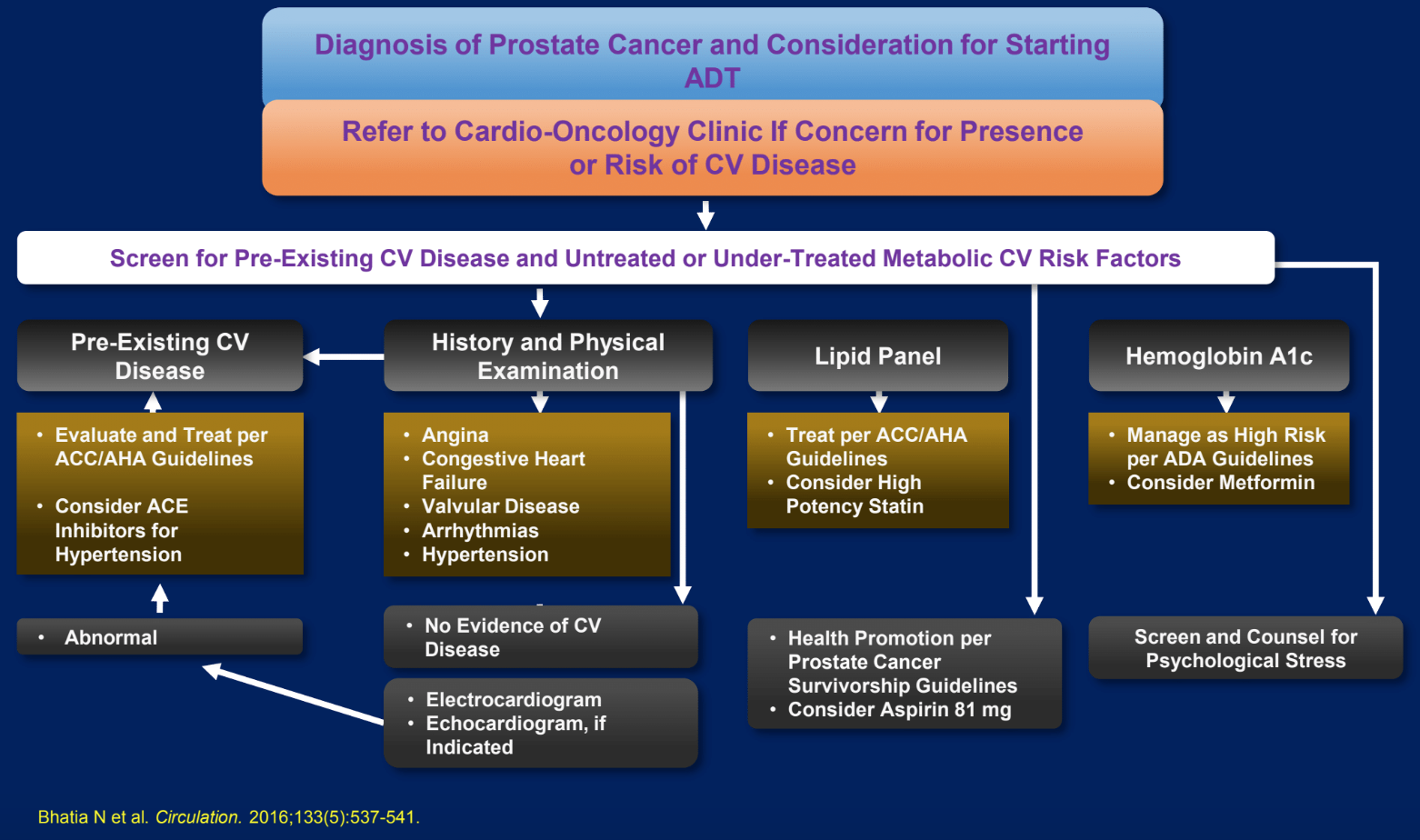
Figure 1. An algorithm developed to address treatment for prostate cancer patients
Heart disease remains a greater threat to many prostate cancer patients than the cancer itself. Dr. Shore concluded that ADT is the mainstay of treatment of advanced prostate cancer in one of the most common cancers, globally. It is imperative that we understand the associated complications of ADT and how to optimize their treatments. It is critical that the primary care physician and cancer care providers work together to prevent ADT complications.
Presented by: Neil Shore, MD, Medical Director for the Carolina Urologic Research Center, Myrtle Beach, South Carolina, USA
Written by: Hanan Goldberg, MD, Urology Department, SUNY Upstate Medical University, Syracuse, New York, USA, Twitter: @GoldbergHanan at the 2019 Advanced Prostate Cancer Consensus Conference (APCCC) #APCCC19, Aug 29 – 31, 2019 in Basel, Switzerland
References:
1. Webb, Carolyn M., John G. McNeill, Christopher S. Hayward, Dominique De Zeigler, and Peter Collins. “Effects of testosterone on coronary vasomotor regulation in men with coronary heart disease.” Circulation 100, no. 16 (1999): 1690-1696.
