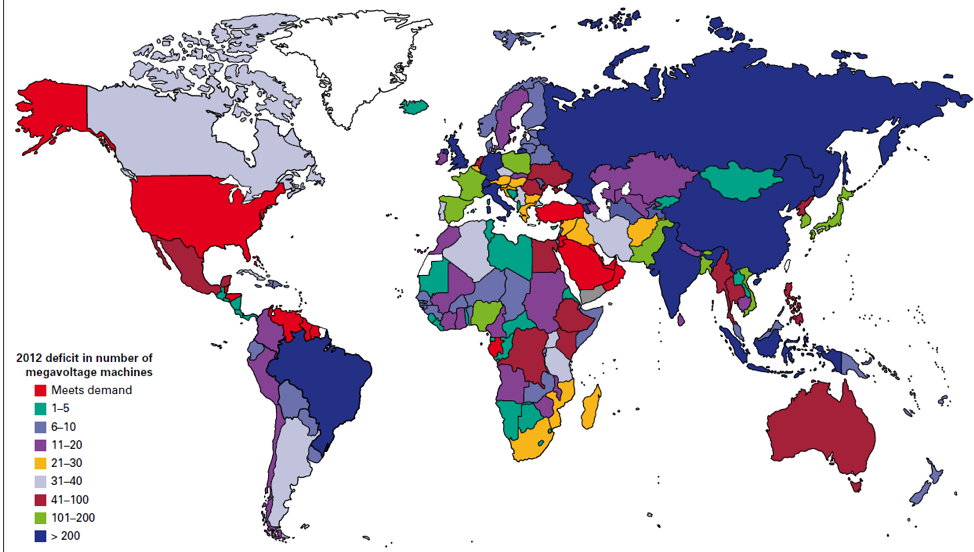
Basel, Switzerland (UroToday.com) Dr. Karim Fizazi gave a brief talk on what can be approved in global access to treatment of advanced prostate cancer. His talk began with data on current access to radiotherapy on a worldwide level (Figure 1). This data showed that there is still a long way to go for us to reach the required numbers in a large percentage of countries in the world.
Figure 1 – Access to radiotherapy worldwide – Difference between demand and supply of radiotherapy machines:
The next topic discussed was the use of generic drugs, which will significantly lower the price needed to pay for the treatment of each patient. Lowering the price significantly will enable to make the medication available to a much larger proportion of the population. An example of an already commonly used generic drug is docetaxel. Before the loss of exclusivity, the cost of docetaxel per one cycle of use was approximately 1500 Euro. When the drug became generic, the cost came down to 20 Euro per cycle! However, according to a recently published paper, the cost of other generic drugs might not be sufficiently low to sustain publicly funded health care systems.1
For abiraterone, the expected access to generic medication is in 2019. In the US the cost is expected to decrease from 9000 USD to 2500 USD for uninsured patients, and no major change is expected for insured patients. In Brazil, the price is expected to go down from 3000 USD to approximately 1300-2300 USD, and in Europe, abiraterone is patent protected until September 2022. However, specifically for abiraterone, there may be an alternative that irrespective of the generic medication issue. A recently published study demonstrated that low-dose abiraterone with food was just as effective as standard dose abiraterone.2 In this study, low dose abiraterone with low-fat breakfast was shown to be non-inferior to standard dosing abiraterone with respect to PSA metrics. Reducing the dose of abiraterone by ¾ is could tremendously reduce the associated cost, and this requires further research.
The next drug discussed was cabazitaxel, with its generic form expected to start in 2021. CARD – is a randomized phase 4 trial comparing cabazitaxel and an androgen receptor (AR)-targeted agent in men with metastatic castration-resistant prostate cancer (mCRPC) progressing after docetaxel and an alternative AR-targeted agent. The results of this trial are expected to be shown at the ESMO meeting in 2019, in Barcelona. This trial will show us if cabazitaxel has the same efficacy as abiraterone for example or better.
For the other drugs used in prostate cancer, the expected loss of patent is as follows:
Denosumab – 2025, enzalutamide – 2027, apalutamide – 2032, with no known estimated dates for Radium-223 and darolutamide.
Another important issue that is sometimes overlooked is the use of surgical castration vs. the use of LHRH agonists and antagonists. Surgical castration is effective as medical castration but is significantly cheaper. In a recently published study, 10,000 prostate cancer metastatic patients were assessed, and predictors of surgical castration were searched for.3 The study showed that the likelihood to receive surgical castration is greatly associated with the following factors:
- Hispanic race (OR 1.3)
- Low socioeconomic status (OR 2)
- Rural area (OR 1.5)
- Medicaid/public insurance (OR 2.2)
In contrast, patients with military/Veterans Affairs insurance were significantly less likely to receive surgical castration compared with patients with private insurance (OR, 0.34; 95% CI, 0.13-0.88). It would seem that low socioeconomic status is generally associated with a greater probability of undergoing surgery.
Dr. Fizazi concluded his talk at the Advanced Prostate Cancer Consensus Conference (APCCC) 2019 hoping that in 2 years’ time, at the APCCC 2021 meeting, the global access to various prostate cancer treatments will be greatly improved.
Presented by: Karim Fizazi, MD, Ph.D. Professor of Medicine of Institut Gustave Roussy, (IGR) in Villejuif, France
Written by: Hanan Goldberg, MD, Urology Department, SUNY Upstate Medical University, Syracuse, New-York, USA @GoldbergHanan at the 2019 Advanced Prostate Cancer Consensus Conference (APCCC) #APCCC19, Aug 29 – 31, 2019 in Basel, Switzerland
References:
- Cheung WY, Kornelsen EA, Mittmann N, et al. The economic impact of the transition from branded to generic oncology drugs. Current oncology (Toronto, Ont.). Apr 2019;26(2):89-93.
- Szmulewitz RZ, Peer CJ, Ibraheem A, et al. Prospective International Randomized Phase II Study of Low-Dose Abiraterone With Food Versus Standard Dose Abiraterone In Castration-Resistant Prostate Cancer. Journal of clinical oncology : official journal of the American Society of Clinical Oncology. May 10 2018;36(14):1389-1395.
- Borno HT, Lichtensztajn DY, Gomez SL, Palmer NR, Ryan CJ. Differential use of medical versus surgical androgen deprivation therapy for patients with metastatic prostate cancer. Cancer. Feb 1 2019;125(3):453-462.
