Basel, Switzerland (UroToday.com) Dr. Christopher Sweeny’s talk encompassed systemic therapy for prostate cancer. The spectrum of patients starting androgen deprivation therapy (ADT) for metastatic disease is quite vast. There are patients who present with de novo metastatic disease, and some present after previous radical prostatectomy or radiotherapy. Some of the patients are fit and young and some are old and frail. Some have a minimal disease, and some have widespread disease seen on conventional imaging modalities. Lastly, some patients have received ADT with radiotherapy or prostatectomy.
Metastatic hormone-sensitive prostate cancer (mHSPC) patients have a variable response to testosterone suppression, as can be seen in the CHAARTED1 and GETUG 15 trials2 (Table 1).
Table 1- Different patients have variable response to testosterone suppression:
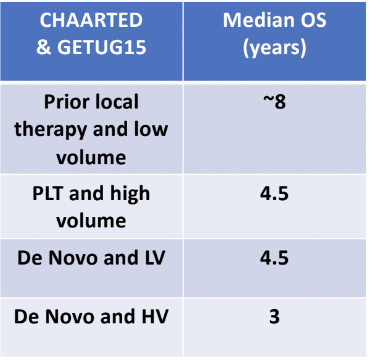
From the long-term follow-up of the CHAARTED study after a median follow-up of 53.7 months, for the entire population, we can still see that those who got docetaxel+ADT continued to benefit compared to those who got ADT alone, with a 10-month survival advantage (HR 0.73).3 Specifically in the low volume metastatic disease population, we can see that the indolent patients dilute long term overall survival with docetaxel and we cannot make definitive statements on interim/early results. A few of the low-volume metastatic disease have aggressive disease and benefit from early docetaxel. Currently, it is not possible to identify them, and they do not constitute a large enough proportion to affect the survival of the entire group. In contrast, the high-volume metastatic disease patients benefited even more from docetaxel after long term follow-up.
When assessing the quality of life of these patients – for the ones that received only ADT, the ones with the low-volume disease had no change in the quality of life over 12 months. However, in high volume metastatic disease patients, the quality of life declined, as the disease had progressed. In contrast, for patients who received both ADT and docetaxel: For patients who received ADT only there was a decline in the quality of life for those with low-volume metastatic disease, but for the ones with high-volume metastatic disease there was no decline in quality of life.4
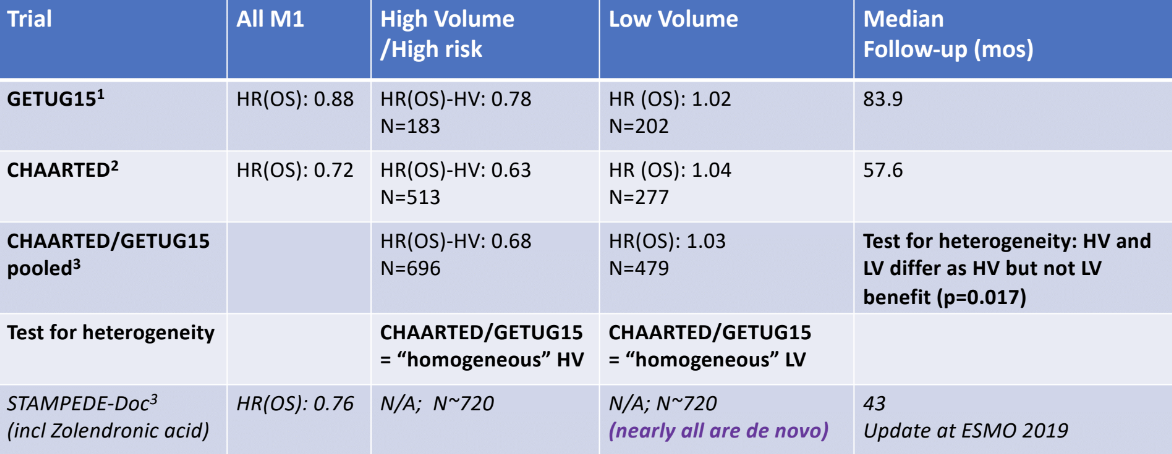
Next, Dr. Sweeney discussed the effect of these treatments on overall survival, as measured by hazard ratio (HR), and can be seen in the summary table 2. Docetaxel has a direct overall survival benefit for high-volume disease and documented improvement in the quality of life. However, for low-volume disease, there appears to be no overall survival benefit for docetaxel. Disease volume appears to be prognostic for the outcome on ADT and predictive for docetaxel benefit. Does this mean that there are different disease biology processes within mHSPC?
Table 2 – Summary of ADT +/- docetaxel treatment effect on overall survival:
The next medication discussed by Dr. Sweeney was abiraterone. The LATITUDE study showed that patients with high-volume metastatic disease (using CHAARTED criteria) benefit from abiraterone (HR 0.62 95% ci 0.52-0.74, P<0.0001), while for low volume disease there appears to be no statistically significant benefit (HR 0.72, 95% CI 0.47-1.10, p=0.1242).5 Abiraterone in mHSPC patients appeared to slow the time to decline in quality of life.6 When looking at the data from the STAMPEDE-abiraterone data, there was also a clear benefit to abiraterone in high volume disease (overall survival difference of 19.7%, HR 0.54, 95% CI 0.41-0.70, p<0.001), and for low volume disease (overall survival difference of 4.4%, HR 0.66, 95% CI 0.44-0.98, p=0.041). We need longer-term overall survival data to see if the overall survival benefit will be greater with the early use of abiraterone.
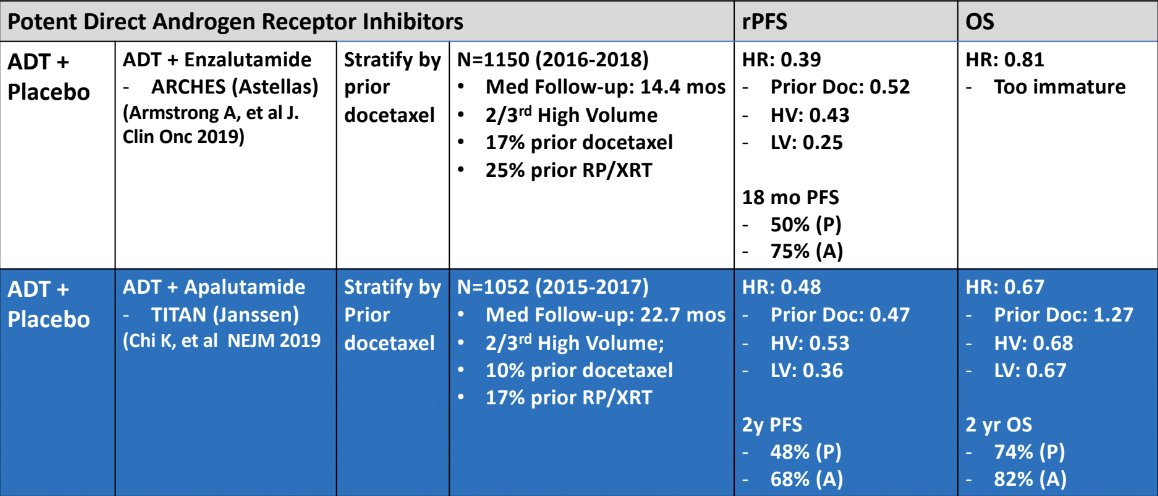
The next medication discussed was enzalutamide. The ENZAMET trial’s primary endpoint was overall survival difference between mHSPC patients treated with enzalutamide and ADT compared to those treated with ADT alone.7 This trial demonstrated a clear benefit for the enzalutamide treated patients (Figure 1). In this trial, there is a mixed bag of patients with low- and high- volume disease, de novo vs. metachronous metastases, and many permutations. Table 3 demonstrates the scorecard of other medications which are potent direct androgen receptor inhibitors in mHSPC. These include apalutamide and enzalutamide in the ARCHES trial.8
Figure 1 – Enzamet primary endpoint of overall survival showing the benefit in those treated with enzalutamide:
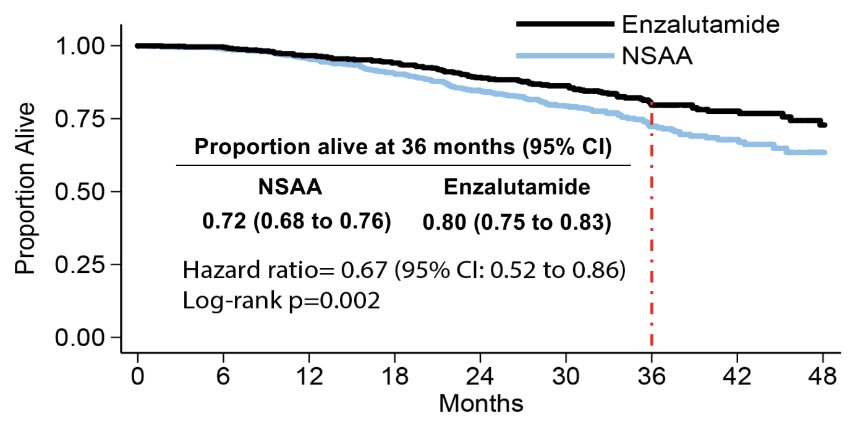
Table 3 – Scorecard of other trials of potent direct androgen receptor inhibitors in mHSPC:
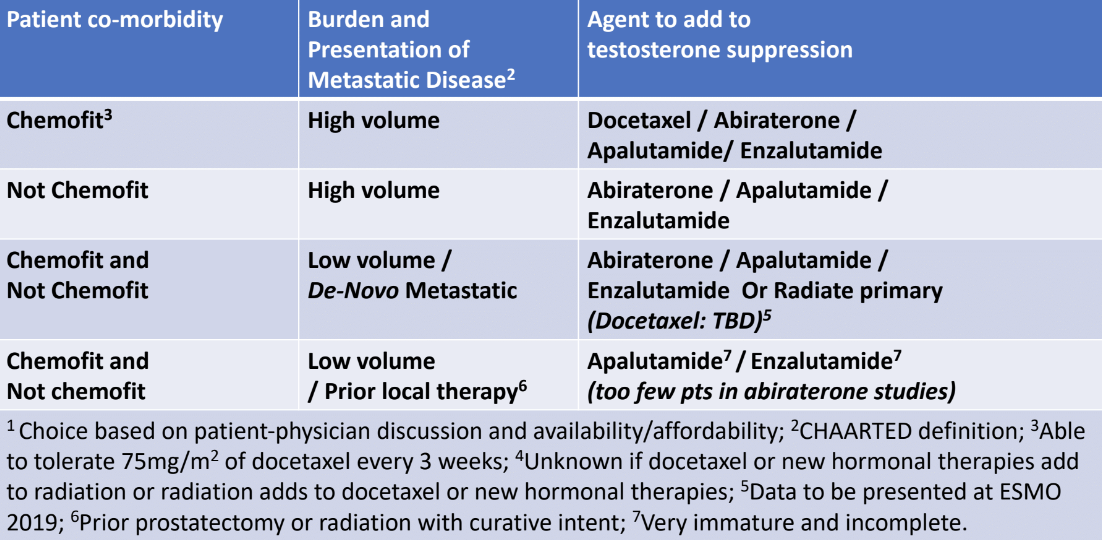
Dr. Sweeney ended his talk with a summary table showing direct evidence of some overall survival treatment benefit in mHSPC patients, as of August 2019 (Table 4). Dr. Sweeney concluded his talk stating that it is important to look for patterns in the available data to make decisions for patients. It is important to incorporate disease burden vs. comorbidities vs. treatment benefit vs. treatment risk. Another important issue to revisit at this point in time is treatment breaks, as over 60% of patients are on abiraterone + prednisone, enzalutamide, or apalutamide for more than 3 years. Lastly, we must find usable biomarkers to help us guide therapy in this developing field of treatment.
Table 4 – Overall survival treatment benefit in mHSPC patient as of August 2019:
Presented by: Christopher Sweeney, MBBS, Professor of Medicine, Harvard Medical School, Medical Oncologist, Dana-Farber Cancer Institute, Boston, Massachusetts, USA
Written by: Hanan Goldberg, MD, Urology Department, SUNY Upstate Medical University, Syracuse, New-York, USA @GoldbergHanan at the 2019 Advanced Prostate Cancer Consensus Conference (APCCC) #APCCC19, Aug 29 – 31, 2019 in Basel, Switzerland
References:
- Sweeney C. et al. Chemohormonal Therapy in Metastatic Hormone-Sensitive Prostate Cancer. New England Journal of Medicine. 2015; 373:737-746
DOI: 10.1056/NEJMoa1503747. - Gravis G. et al. Androgen-deprivation therapy alone or with docetaxel in non-castrate metastatic prostate cancer (GETUG-AFU 15): a randomised, open-label, phase 3 trial. Lancet Oncology. 2013 Feb;14(2):149-58. doi: 10.1016/S1470-2045(12)70560-0.
- Kyriakopolous et al. Chemohormonal Therapy in Metastatic Hormone-Sensitive Prostate Cancer: Long-Term Survival Analysis of the Randomized Phase III E3805 CHAARTED Trial. Journal of Clinical Oncology. 2018 DOI: 10.1200/JCO.2017.75.3657.
- Morgans et al. Quality of Life During Treatment With Chemohormonal Therapy: Analysis of E3805 Chemohormonal Androgen Ablation Randomized Trial in Prostate Cancer. Journal of Clinical Oncology. 2018 Apr 10;36(11):1088-1095. doi: 10.1200/JCO.2017.75.3335.
- Fizazi K. et al. Abiraterone plus Prednisone in Metastatic, Castration-Sensitive Prostate Cancer. New England Journal of Medicine. 2017; 377:352-360
DOI: 10.1056/NEJMoa1704174. - Chi K. et al. Patient-reported outcomes following abiraterone acetate plus prednisone added to androgen deprivation therapy in patients with newly diagnosed metastatic castration-naive prostate cancer (LATITUDE): an international, randomised phase 3 trial. Lancet Oncology. Feb;19(2):194-206. doi: 10.1016/S1470-2045(17)30911-7. 2018.
- Sweeney C. et al. Enzalutamide with Standard First-Line Therapy in Metastatic Prostate Cancer. New England Journal of Medicine. 2019; 381:121-131
DOI: 10.1056/NEJMoa1903835. - Armstrong A. et al. ARCHES: A Randomized, Phase III Study of Androgen Deprivation Therapy With Enzalutamide or Placebo in Men With Metastatic Hormone-Sensitive Prostate Cancer. Journal of Clinical Oncology. 2019 DOI: 10.1200/JCO.19.00799.
