Barcelona, Spain (UroToday.com) There are three randomized trials, RADICALS (ISRCTN40814031),1 GETUG-AFU 17 (NCT00667069) and RAVES (NCT00860652); recently presented at ASTRO 2019, that have compared adjuvant radiotherapy with a policy of salvage radiotherapy for PSA failure after radical prostatectomy for men with localized prostate cancer. However, these trials have limited power for long-term outcomes. At the ESMO 2019 prostate cancer session, Claire Vale presented initial results of a pre-planned meta-analysis of the above trials. The ARTISTIC collaboration prospectively planned a series of meta-analyses for each outcome.
Initial discussions for a meta-analysis occurred in 2011 with the RADICALS-RT and RAVES group. In 2014, the ARTISTIC collaboration was officially launched. The authors used a framework for adaptive meta-analysis (FAME), prospectively defining their methods, including a consistent definition of PSA-driven event-free survival, prior to knowledge of trial results (CRD42019132669). Event-free survival was defined as time from randomization to any of (i) PSA >=0.4 ng/mL and rising after completion of radiotherapy, (ii) clinical/radiological progression, (iii) initiation of non-trial treatment, (iv) death from prostate cancer following completion of radiotherapy, or (v) PSA level >= 2.0 ng/mL at any time after randomization. They anticipated 240 events across all trials by Fall 2019, giving 90% power to detect a 5% absolute difference in 5-year event-free survival.
Across the 3 trials, 1,074 men were randomized to adjuvant radiotherapy and 1,077 to salvage radiotherapy. The trials are comparable, but with subtleties noted below:
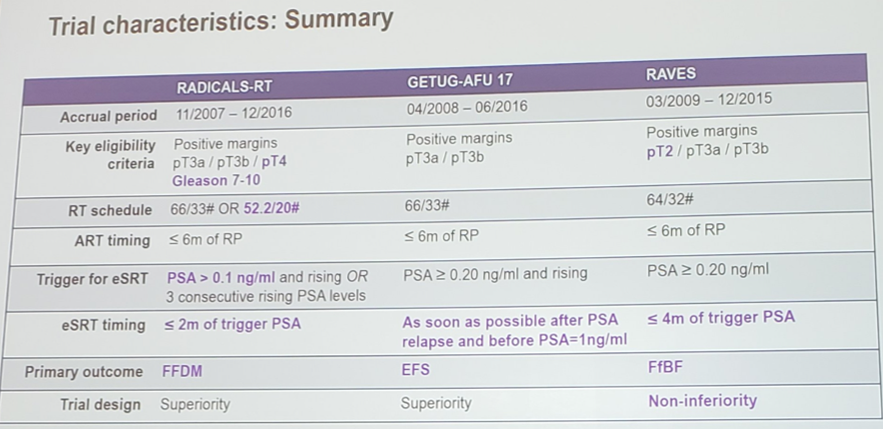
To date, 395 men (37%) had commenced salvage radiotherapy. Patient characteristics were balanced within trials and overall. Men had a median age of 65 years and most (77%) had a Gleason sum score of 7. Median follow-up ranged from 47 to 61 months. In August 2019, RADICALS and GETUG-AFU 17 provided event-free survival results for the meta-analysis (interim for GETUG-AFU 17). RAVES currently could only supply freedom from biochemical failure results. However, as the vast majority of first events across all trials are biochemical failures, these results were pooled in a preliminary meta-analysis of event-free survival. Based on 245 events, the meta-analysis shows no evidence that event-free survival is improved with adjuvant radiotherapy compared to salvage radiotherapy (HR 1.09, 95% CI 0.86-1.39, p = 0.47).
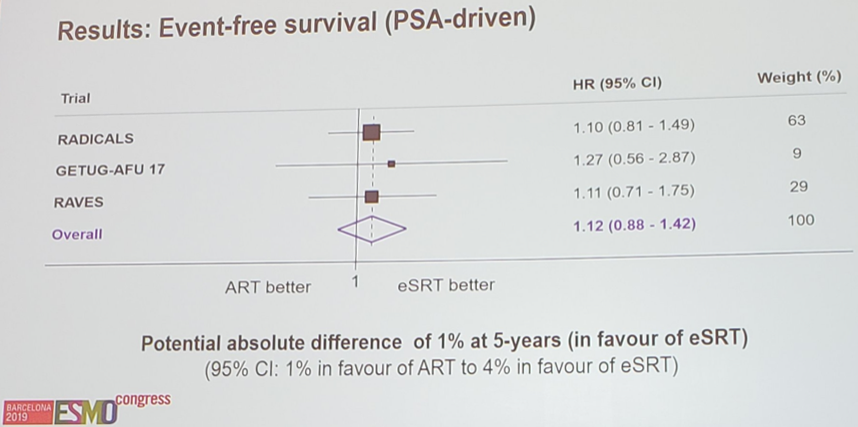
Professor Vale concluded that this collaborative, prospective and early meta-analysis of all men from three randomized trials, suggests that salvage and adjuvant radiotherapy offer similar outcomes for event-free survival. Importantly, salvage radiotherapy spares many men from receiving radiotherapy, and associated side-effects. Final data from GETUG-AFU 17 and RAVES may help establish whether some subgroups of men might benefit from either treatment. Finally, longer follow-up is needed for a meta-analysis of metastasis-free survival. The plan for the current analysis is to publish the results simultaneously to the three trials.
Clinical trial identification
ISRCTN40814031; NCT00667069; NCT00860652
Presented by: Claire Vale, Ph.D., MRC Clinical Trials Unit, University College London, Institute of Clinical Trials and Methodology-UCL, United Kingdom
Co-authors: M. Brihoum,2 S. Chabaud,3 A. Cook,1 D. Fisher,1 S. Forcat,4 C. Fraser-Browne,5 A. Herschtal,6 A. Kneebone,7 S. Nénan,2 C. Parker,8 M. Parmar,9 M. Pearse,5 P. Richaud,10 E. Rogozińska,4 P. Sargos,11 M. Sydes,9 J. Tierney1
1. Institute of Clinical Trials and Methodology-UCL, London, UK
2. Unicancer, Paris, FR
3. Centre Léon Bérard, Lyon, FR
4. Institute of Clinical Trials and Methodology, UCL, London, UK
5. Auckland City Hospital, Auckland, NZ
6. Peter MacCallum Cancer Centre, Melbourne, AU
7. Northern Sydney Cancer Centre, Sydney, AU
8. The Institute of Cancer Research/Royal Marsden NHS Foundation Trust, Sutton, UK
9. MRC Clinical Trials Unit at UCL, London, UK
10. Institut Bergonie, Bordeaux, FR
11. Institute Bergonié, Bordeaux, FR
Written by: Zachary Klaassen, MD, MSc – Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia Twitter: @zklaassen_md at the 2019 European Society for Medical Oncology annual meeting, ESMO 2019 #ESMO19, 27 Sept – 1 Oct 2019 in Barcelona, Spain
