Barcelona, Spain (UroToday.com) Altered glucose and glutamine metabolism is a hallmark of renal cell carcinoma (RCC). Abnormal glucose metabolism, known as the Warburg effect, deprives the citric acid cycle (TCA cycle) of critical metabolites. Cancer cells compensate for the Warburg effect by increasing glutamine metabolism to sustain the TCA cycle for growth and proliferation. Glutaminase is a key enzyme in glutamine metabolism and drives proliferation of RCC cells when overexpressed. Telaglenastat is a novel, first-in-clinic, selective glutaminase inhibitor that blocks critical glutamine-dependent pathways and synergizes preclinically with signal transduction inhibitors (eg, everolimus). In a previous phase Ib study in metastatic (mRCC), telaglenastat + everolimus was well tolerated and had encouraging clinical activity – 92% disease control rate and 5.8-month median progression free survival (PFS). During the non-prostate cancer session at the 2019 European Society for Medical Oncology annual meeting (ESMO), Dr. Lee and colleagues presented results of ENTRATA, a randomized phase II study of telaglenastat + everolimus vs. placebo + everolimus in heavily pre-treated mRCC patients.
Eligible patients for ENTRATA had ≥2 prior lines of systemic therapy for mRCC, including ≥1 VEGFR-targeted tyrosine kinase inhibitor, a Karnofsky performance status of ≥70%, and measurable disease (RECIST 1.1). Patients were stratified by prior lines of tyrosine kinase inhibitors and MSKCC risk; they were then randomized 2:1 to receive telaglenastat (800 mg PO BID) or placebo, plus everolimus (10 mg PO QD), until disease progression/unacceptable toxicity. The primary endpoint was investigator-assessed PFS (RECIST 1.1; 1-sided alpha <0.2).
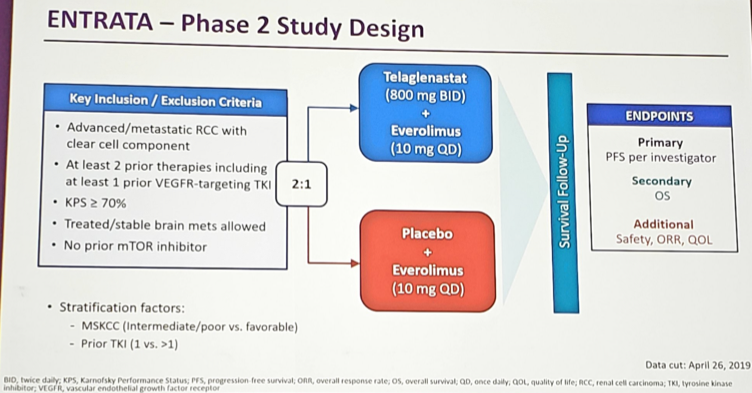
There were 69 patients randomized, including 46 to telaglenastat + everolimus and 23 to placebo + everolimus. Approximately 2/3 were intermediate/poor risk, ~90% had received prior immunotherapy, and the median number of prior treatments was 3. The median PFS was 3.8 months for telaglenastat + everolimus compared to 1.9 months for placebo + everolimus (HR 0.64, 95% CI 0.34-1.20, 1-sided p = 0.079).
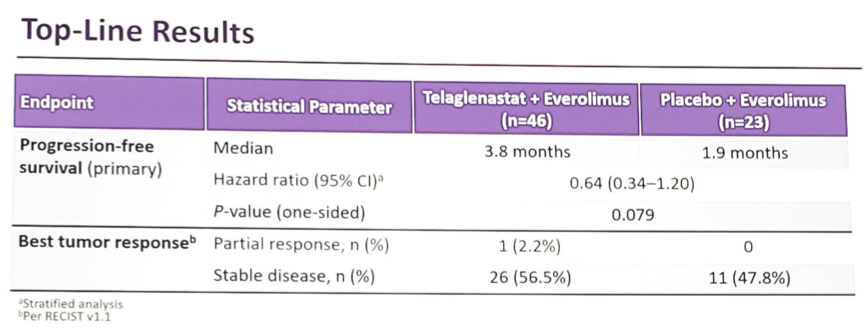
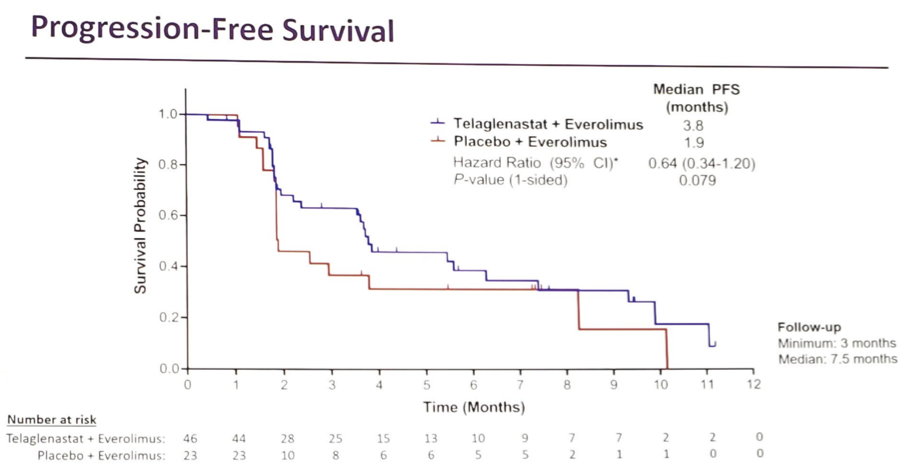
Subgroup analyses were consistent with the primary analysis. Grade ≥3 adverse events occurred in 80% of telaglenastat + everolimus patients vs 60% of placebo + everolimus patients. The most common adverse events were anemia (17% vs 17%), pneumonia (7% vs 4%), abdominal pain (7% vs 0%), thrombocytopenia (7% vs 0%), and fatigue (4% vs 9%). Discontinuation rates due to adverse events were similar between the groups (28% telaglenastat + everolimus, 30% placebo + everolimus), and there were no treatment-related deaths.
Dr. Lee concluded with several points from ENTRATA:
- The addition of telaglenastat improved PFS over placebo + everolimus, with a tolerable safety profile in heavily treated mRCC patients, including refractory to multiple TKIs and immune checkpoint inhibitors
- Data for the secondary endpoint of OS is not yet mature
- ENTRATA met its primary endpoint, supporting proof of concept for glutamine inhibition with telaglenastat as a new therapeutic approach in RCC.
Clinical trial identification
Presented by: Chung-Han Lee, MD, PhD, Medical Oncologist, on behalf of Robert J. Motzer, MD, Medical Oncologist, Kidney Cancer Section Head, Genitourinary Oncology Service; Jack and Dorothy Byrne Chair in Clinical Oncology, Memorial Sloan-Kettering Cancer Center, New York, NY
Co-Authors: C.-H. Lee 1, H. Emamekhoo 2, M. Matrana 3, I. Percent 4, J. Hsieh 5, A. Hussain 6, U. Vaishampayan 7, R. Graham 8, S. Liu 9, S. Mccune 10, M. Shaheen 11, H. Parmar 12, Y. Shen 12, S. Whiting 12, N. Tannir 13
1. Memorial Sloan-Kettering Cancer Center, New York, US
2. Cleveland Clinic, Cleveland, US
3. Ochsner Cancer Institute, New Orleans, US
4. Florida Cancer Specialists – South, Fort Myers, US
5. Washington University School of Medicine in St. Louis, St. Louis, US
6. University of Maryland Greenebaum Comprehensive Cancer Center, Baltimore, US
7. Karmanos Cancer Institute, Detroit, US
8. UT/Erlanger Oncology & Hematology, Chattanooga, US
9. UCLA, Los Angeles, US
10. Northwest Georgia Oncology Centers, Marietta, U
11. The University of Arizona Cancer Center, Tucson, US
12. Calithera Biosciences, South San Francisco, US
13. MD Anderson Cancer Center, Houston, US
Written by: Zachary Klaassen, MD, MSc – Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, Twitter: @zklaassen_md at the 2019 European Society for Medical Oncology annual meeting, ESMO 2019 #ESMO19, 27 Sept – 1 Oct 2019 in Barcelona, Spain
