Barcelona, Spain (UroToday.com) Platinum-based chemotherapy is the standard of care in patients with locally advanced or metastatic urothelial carcinoma, but this therapy is often toxic with overall response rates of 50% or less. The PD-L1 inhibitor Durvalumab was approved by the United States FDA in May 2017 for post-platinum disease based on a phase 1/2 trial showing an 18% objective response rate and 55% one-year overall survival rate. Recent studies have suggested multiple promising biological targets for therapeutic study in advanced urothelial cancer, including FGFR3 mutation or FGFR gene fusion, mTOR pathway alterations such as RICTOR amplification or TSC 1/2 mutations, and DNA damage repair pathways. As reported in this presentation, the BISCAY trial used an innovative design to combine durvalumab immunotherapy with biologically promising targeted approaches available from the AstraZeneca drug development pipeline in attempt to maximize therapeutic outcomes.
Using Foundation One tumor genomic profiling, a total of 391 platinum-refractory patients were screened for pre-specified FGFR (targeted with AZD4547), mTOR (targeted with vistusertib) or DNA damage repair (targeted with olaparib) biomarkers. Treatment arms are shown below. A cohort of patients were allocated durvalumab monotherapy arm (not randomized) to provide a benchmark. The primary endpoints were treatment safety and tolerability. Secondary endpoints were focused on durvalumab efficacy alone or in combination with other therapy. In order to justify further study and identify practice-changing effects, the authors felt they needed a marked improvement with combination therapy over durvalumab alone. They, therefore, powered the study to require 20 patients per arm in order to give a 76% probability of detecting an ORR of 55% of greater. A total of 14-29 patients were allocated to each arm of the study.
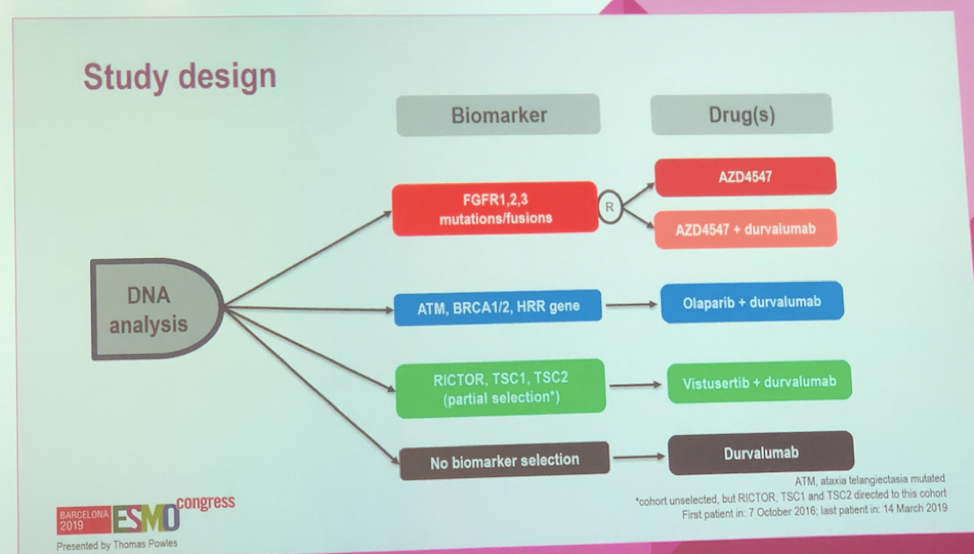
While the cohorts were well-balanced by Bellmunt risk factors, they had differences in PD-L1 status. This is important given the known increased efficacy of durvalumab alone in PD-L1 high patients relative to PD-L1 low tumors. None of the therapeutic arms reached the pre-specified overall response rate cutoff. Combination therapy regimens displayed higher rates of adverse effects, including higher rates of patients for whom adverse events led to treatment discontinuation (33-38% for combination regimens, 20% for AZD4547 alone, 10% durvalumab alone).
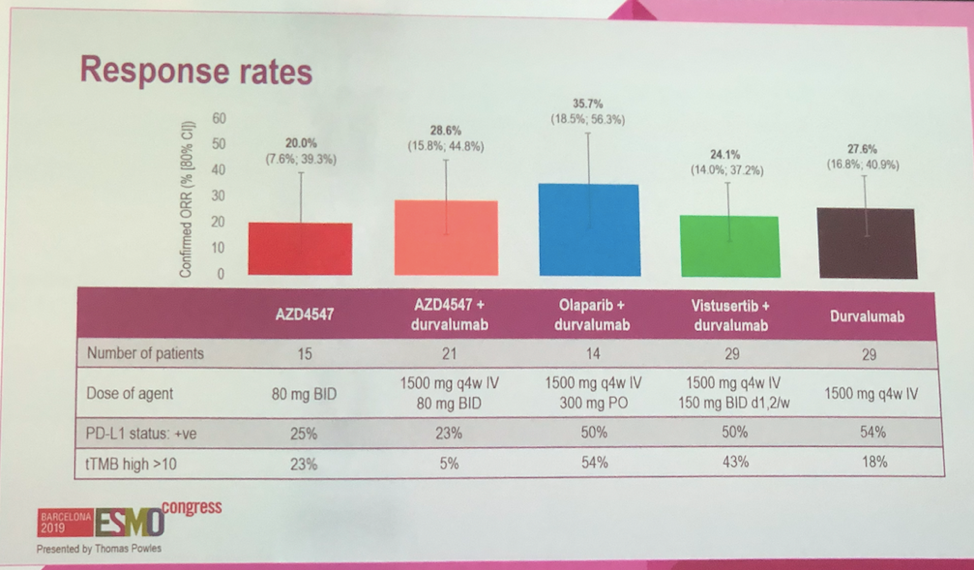
Though no treatment arm reached the pre-specified outcome threshold, clinical activity was seen with biologically-selected combination therapy over durvalumab alone. This comparison is limited by the non-randomized assignment of patients to the durvalumab arm. Adverse event profiles were similar to historical controls. Further trials may explore the efficacy of the biologically-targeted combinations utilized in this study on a larger scale. Importantly, this trial design validates the ability to do small-scale trials using tumor profiling and multiple rationally selected targeted combination therapies and could serve as a model for future studies.
Presented by: Thomas Powles, MBBS, MRCP, MD, Professor of Genitourinary Oncology, Lead for Solid Tumour Research at Barts Cancer Institute, Director of Barts Cancer Institute, London United Kingdom
Written by: Alok Tewari, MD, PhD, Medical Oncology Fellow at the Dana-Farber Cancer Institute, at the 2019 European Society for Medical Oncology annual meeting, ESMO 2019 #ESMO19, 27 Sept – 1 Oct 2019 in Barcelona, Spain
