Barcelona, Spain (UroToday.com) There has been much interest in improving disease free survival (DFS) and overall survival (OS) among patients with locally advanced kidney cancer undergoing radical nephrectomy. As such, several adjuvant therapy trials for high risk patients have been reported over the last few years. However, unfortunately no trial to date has demonstrated an OS benefit and only one reporting a DFS benefit (S-TRAC). At the ESMO 2019 annual congress, Tim Eisen, Ph.D., presented results of SORCE, a randomized, double-blind trial of sorafenib after surgical excision of primary renal cell carcinoma (RCC) at intermediate or high risk of recurrence based on Leibovich classification.
SORCE recruited patients from 147 sites in the UK, Australia, France, Belgium, Denmark, the Netherlands and Spain and randomized patients 2:3:3 between three years of placebo, one year of sorafenib followed by two years of placebo, and three years of sorafenib. The initial sorafenib dose was 400mg twice per day orally, amended during trial recruitment to a reduced starting dose of 400mg daily. The primary outcome was investigator-reported DFS. Given the results of the ASSURE and S-TRAC trials, and blinded to SORCE outcomes, the investigators revised the primary analysis to compare three years of sorafenib vs placebo to focus on the question of longer exposure to sorafenib.
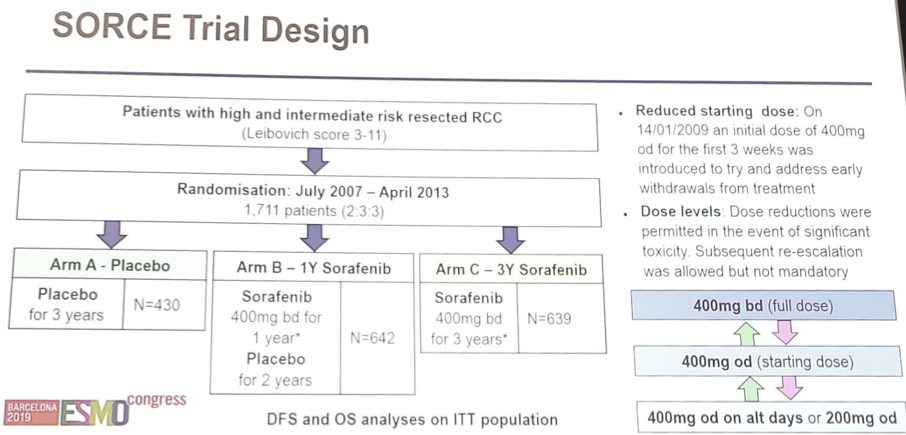
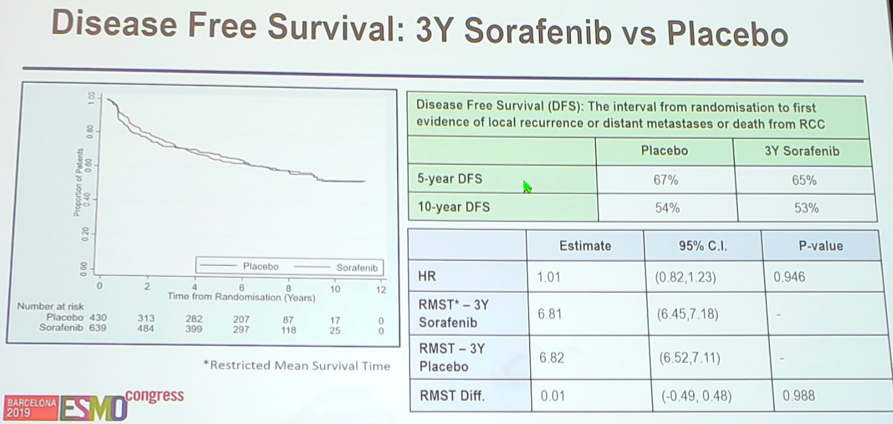
Between July 2007 and April 2013, 1,711 patients were randomized to placebo (n=430), 1-year of sorafenib (n=642), and 3-years of sorafenib (n=639). Mean age was 58 years (range 19-86 year), 71% male, 84% had clear cell histology, and 53% were at intermediate risk of recurrence, whereas 47% were at high risk of recurrence. There were no differences in DFS or OS for all randomized patients, high-risk patients only, and patients with only clear cell RCC. Median DFS was not reached for three years of sorafenib or for placebo (HR 1.01, 95% CI 0.82 -1.23, p=0.946). The restricted mean disease free survival time was 6.81 years for three years of sorafenib and 6.82 years for placebo, a difference of 0.01 (95% CI 0.49 – 0.48, p=0.99).
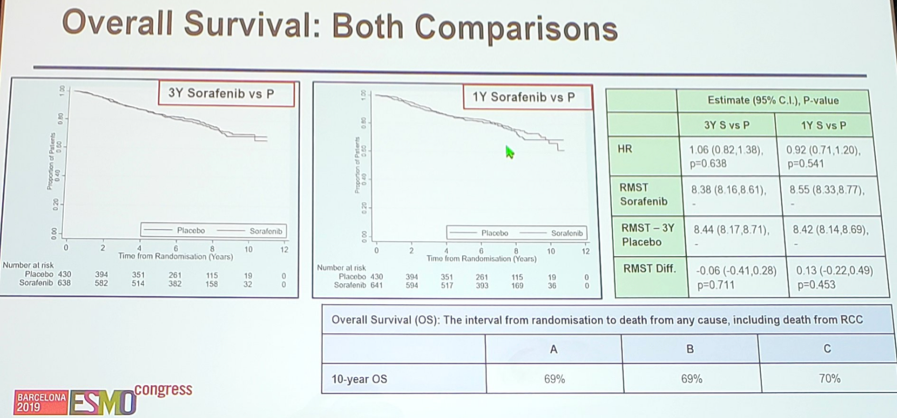
With regards to OS, there was no difference for patients receiving 1 year of sorafenib vs placebo (HR 0.92, 95% CI 0.71-1.20), nor for 3 years of sorafenib vs placebo (HR 1.06, 95% CI 0.82-1.38). The 10-year OS rate for all three groups was 69%-70% — as is shown in the tables below.
Despite offering treatment adaptations, over half of patients stopped treatment early. Grade 3 hand-foot syndrome was reported in 24% of patients on sorafenib.
The conclusions of SORCE are as follows:
- Sorafenib should not be used as adjuvant therapy for RCC.
- Active surveillance remains the standard of care for patients at intermediate or high risk of recurrence following nephrectomy.
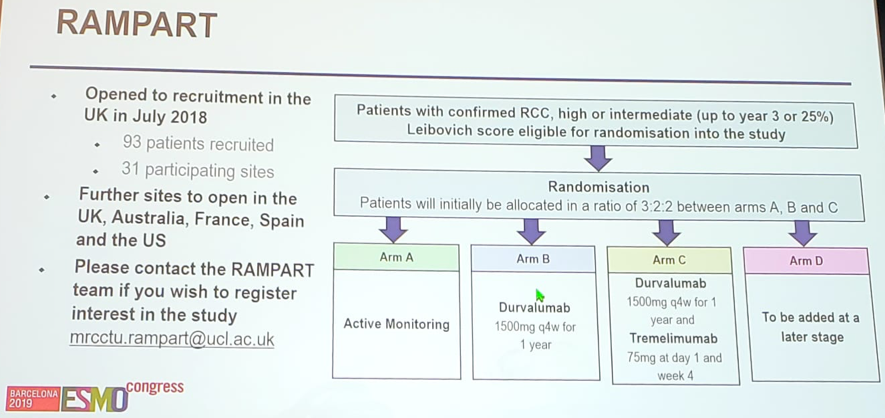
- Surveillance is the appropriate control of the current international adjuvant RCC trial, RAMPART – a multi-arm trial assessing check point inhibitors as adjuvant therapy.
Clinical trial information: ISRCTN38934710; EudraCT: 2006-006079-19; NCT00492258
Presented by: Tim Q. Eisen, Ph.D., Cambridge University Hospitals NHS Foundation Trust, Addenbrooke’s Hospital, Cambridge, United Kingdom
Written by: Zachary Klaassen, MD, MSc – Assistant Professor of Urology, Georgia Cancer Center, Augusta University/Medical College of Georgia, @zklaassen_md, at the 2019 European Society for Medical Oncology annual meeting, ESMO 2019 #ESMO19, 27 Sept – 1 Oct 2019 in Barcelona, Spain
