Barcelona, Spain (UroToday.com) PD-1 inhibition can lead to deep, durable responses in metastatic castration-resistant prostate cancer (mCRPC), but only in a minority of patients. Graff and colleagues previously reported five (18%) responders among 28 patients receiving pembrolizumab with enzalutamide, however, no marker has been shown to universally predict response. Analysis of fecal microbiota from patients with epithelial cancers previously showed an association between elevated Akkermansia muciniphila (A. muc) levels and response to PD-1 inhibitors. During the prostate cancer poster session at ESMO 2019, Graff and colleagues presented data of an additional 30 patients obtaining tumor biopsies, blood, and fecal specimens collected for analysis.
The 30 patients with mCRPC progressing on enzalutamide received pembrolizumab 200 mg IV every 3 weeks for 4 doses with continued enzalutamide. Prior chemotherapy for mCRPC was prohibited. The primary endpoint for this expansion study was the proportion of patients with a prostate-specific antigen (PSA) decline of ≥ 50%. The secondary endpoints were objective response, PSA progression-free survival (PFS), radiographic PFS, overall survival (OS) and correlative studies.
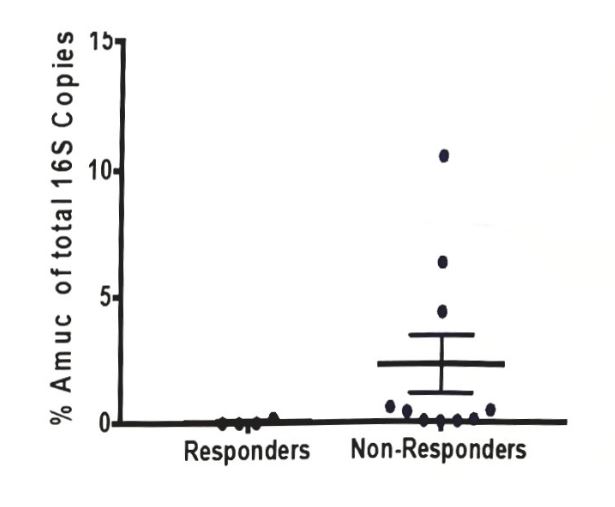
Four of 30 (13%) patients achieved a PSA reduction ≥ 50%, and six of 24 (25%) patients with measurable disease responded radiographically. After a median follow-up of 17.4 months, median PSA PFS was 5.6 months (95%CI 3.5-8.1) and median OS was 17.3 month (95% CI 7.7-17.7). Of 27 patients evaluable for radiographic PFS, 17 (63%) have progressed, the median radiographic PFS was 5.3 months (95% CI 2.6-11.2), and eight patients had immune-related adverse events. Single-cell transcriptomic analysis prior to PD-1 treatment in 12 patients revealed enrichment in a CD8+ exhausted T cell population (p < 0.0003) in patients who responded to PD-1. Among the 30 patients enrolled, 14 patients provided a baseline fecal sample. Interestingly, responders by PSA and/or imaging had a significantly lower abundance of A. muc by quantitative PCR (p < 0.022).
The authors concluded:
- The expansion cohort validates the prior results that pembrolizumab has activity in enzalutamide-resistant patients
- Using single-cell RNA sequencing, the authors revealed a T-cell signature associated with a response, an observation not possible at the bulk transcriptome level
- Responders had lower levels of fecal A. muc than non-responders, suggesting markers of PD-1 response in mCRPC may be different than in other cancer types such as melanoma
Clinical trial identification: NCT02312557.
Presented by: Julie N. Graff, MD, OHSU Knight Cancer Institute, Portland, Oregon
Co-Authors: A. Moran,2 R. Slottke,1 G. Thomas,2 J. Alumkal,2 R. Thompson,2 M. Wood,2 L. Peiffer, 3 R. Bergan,2 K. Sfanos,3 T. Beer2
1. OHSU Knight Cancer Institute, Portland, Oregon
2. Oregon Health & Science University, Portland, Oregon
3. Johns Hopkins University, Baltimore, Maryland
