Athens, Greece (Urotoday.com) Dr. Grivas gave a talk supporting the role of neoadjuvant chemotherapy (NAC) in muscle-invasive bladder cancer (MIBC). This was part of a debate with Dr. Mark Soloway, who later gave a talk supporting the role of adjuvant chemotherapy in MIBC.
The rationale for NAC entails several important points. First, it is an earlier attempt to eradicate micrometastases, which can be the most common cause of cancer-related morbidity and mortality. Additionally, it can potentially downstage the bladder tumor, resulting in a higher rate of complete response and better outcomes. The advantage of NAC is that patients can get it immediately and they do not need to recover after surgery if they chose adjuvant chemotherapy instead. NAC gives us the opportunity to assess tumor biology and behavior in vivo in real-time, having substantial implications on overall management. It also allows us to interrogate biomarkers in tumor tissue, blood, urine, and stool for research. Lastly and most importantly, we have level one evidence supported by phase 3 trials showing the beneficial role of NAC. It has also been adopted by all guidelines.
The SWOG 8710 trial1 demonstrated a 30-month improvement in median overall survival with NAC and radical cystectomy compared to cystectomy alone. This trial showed a clear pathologic complete response, recurrence-free survival and overall survival benefit with NAC and radical cystectomy. There was no higher toxicity or perioperative complications despite the use of an older chemotherapeutic regimen. Another important trial is the EORTC-MRC trial2 which also showed an overall survival benefit for NAC and included clinical stage T2 patients as well.
A large meta-analysis published in Lancet included 10 randomized trials with 2,688 patients and compared cisplatin-based combination NAC plus local therapy vs. definitive local therapy alone.3 The meta-analysis showed a 5-year overall survival of 50% vs. 45% (HR 0.87, 95% CI .78-0.98) in favor of the NAC arm. There was a lower recurrence risk for patients with NAC, translating into an absolute disease-free survival benefit of 7%.
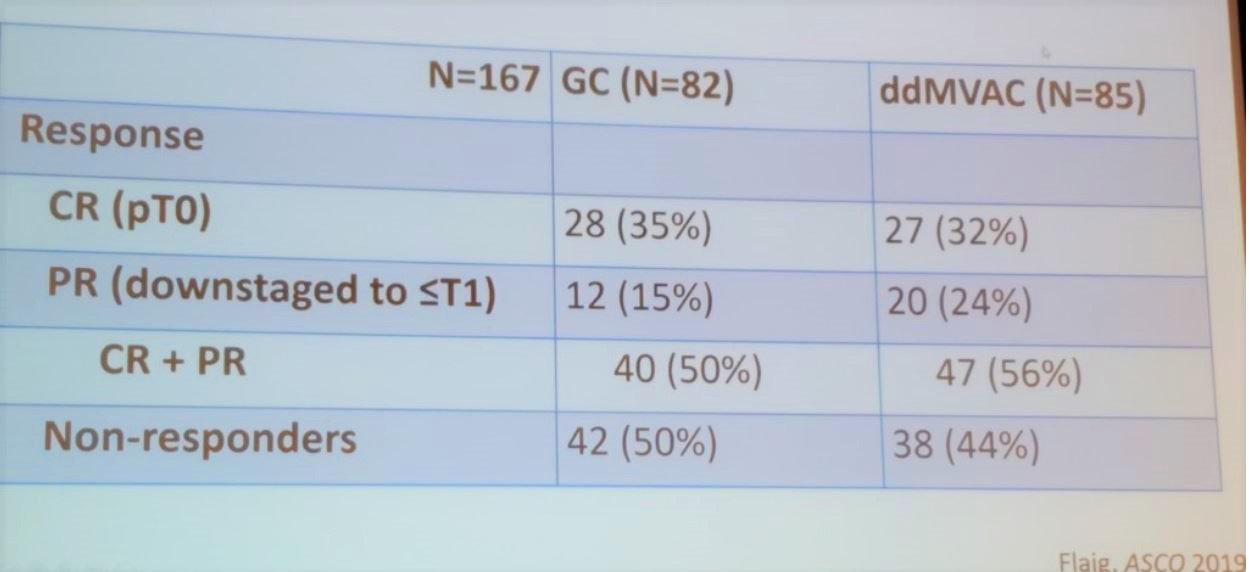
The COXEN trial compared pathologic response by stratifying patients to different NAC regimens. These included gemcitabine + cisplatin, vs. dose dense MVAC (Figure 1 and Table 1).
Figure 1 -COXEN trial design:
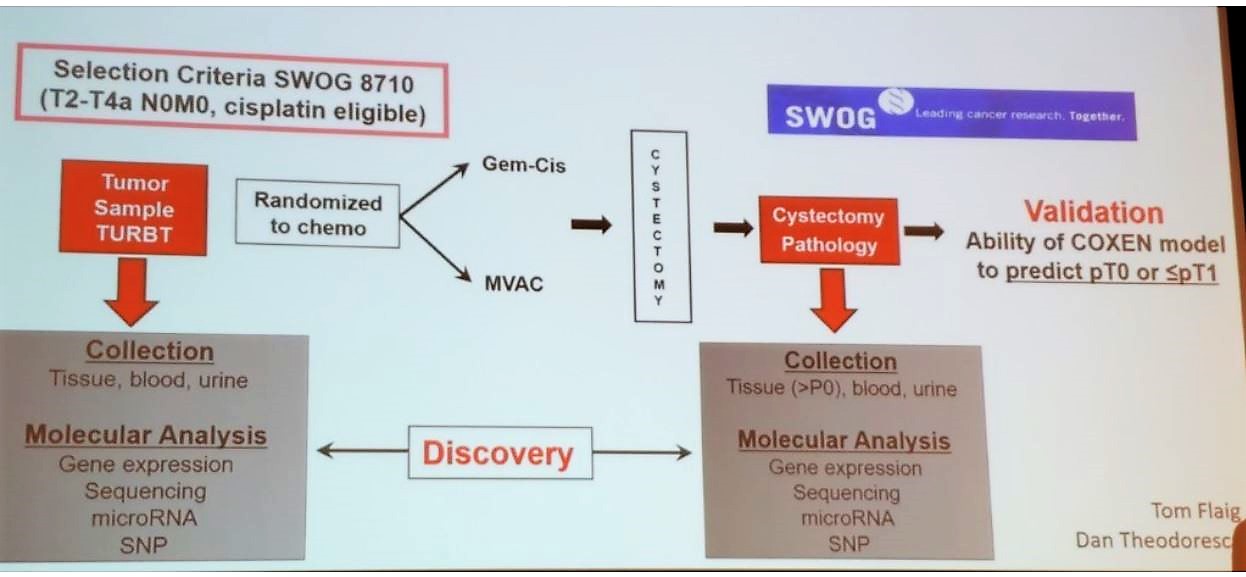
Table 1- COXEN trial results:
Dr. Grivas continued his talk mentioning that inaccurate staging is quite common in bladder cancer patients. 54% of clinical T2 patients were shown to be upstaged at the time of radical cystectomy (when not receiving NAC).4
The only comparison between NAC and adjuvant chemotherapy was published recently.5 However, this was a retrospective analysis with all it’s associated biases and limitations. The RISC database was used with 656 MIBC patients analyzed. The median disease-free survival was 34.5 months (95% CI 25.3-43.9) in NAC vs. 24.9 months (95% CI 19.4-30.5) in adjuvant chemotherapy; with 21% lower risk of progression favoring NAC (HR 0.78, 95% CI 0.63-0.96, p=0.02). No significant differences in cancer-specific survival and overall survival were shown. However, again, we need to consider the selection and confounding biases in this retrospective analysis, when assessing these results.
Dr. Grivas concluded his talk reiterating some important points. NAC improves pathologic complete response rates, recurrence-free survival, and overall survival. NAC is currently a routine practice, which is overall safe, with a manageable side effect profile, and with no negative impact on surgical morbidity or mortality. To date, there is no molecular biomarker with proven clinical utility, and until molecular profiling may enable better patient selection, we should continue to follow the current standard of care, and treat patients with NAC before radical cystectomy.
Presented by: Petros Grivas, MD, PhD, Clinical Director of the Genitourinary Cancers Program, University of Washington, Seattle, Washington
Written by: Hanan Goldberg, MD, Urology Department, SUNY Upstate Medical University, Syracuse, New York, USA, Twitter: @GoldbergHanan at the 39th Congress of the Société Internationale d’Urologie, SIU 2019, #SIUWorld #SIU2019, October 17-20, 2019, Athens, Greece
References:
1. Grossman HB, Natale RB, Tangen CM, et al. Neoadjuvant Chemotherapy plus Cystectomy Compared with Cystectomy Alone for Locally Advanced Bladder Cancer. New England Journal of Medicine 2003; 349(9): 859-66.
2. Griffiths G, Hall R, Sylvester R, Raghavan D, Parmar MK. International phase III trial assessing neoadjuvant cisplatin, methotrexate, and vinblastine chemotherapy for muscle-invasive bladder cancer: long-term results of the BA06 30894 trial. Journal of clinical oncology : official journal of the American Society of Clinical Oncology 2011; 29(16): 2171-7.
3. Neoadjuvant chemotherapy in invasive bladder cancer: a systematic review and meta-analysis. Lancet (London, England) 2003; 361(9373): 1927-34.
4. Svatek RS, Shariat SF, Novara G, et al. Discrepancy between clinical and pathological stage: external validation of the impact on prognosis in an international radical cystectomy cohort. BJU international 2011; 107(6): 898-904.
5. Del Bene G, Calabro F, Giannarelli D, et al. Neoadjuvant vs. Adjuvant Chemotherapy in Muscle Invasive Bladder Cancer (MIBC): Analysis From the RISC Database. Front Oncol 2018; 8: 463
